Friday, Mar. 23, 2018
The Show Ponies "Kalamazoo"
(~5:45), I'm With Her (Sara Watkins, Sarah Jarosz, Aoife
O'Donovan) "Ring
Them Bells" (3:46), "Little Lies"
(3:51), "Ain't
That Fine" (3:08), Paul Simon & Art Garfunkel "American Tune"
(4:25), Crooked Still (w/ Aiofe O'Donovan) "American Tune"
(3:24)
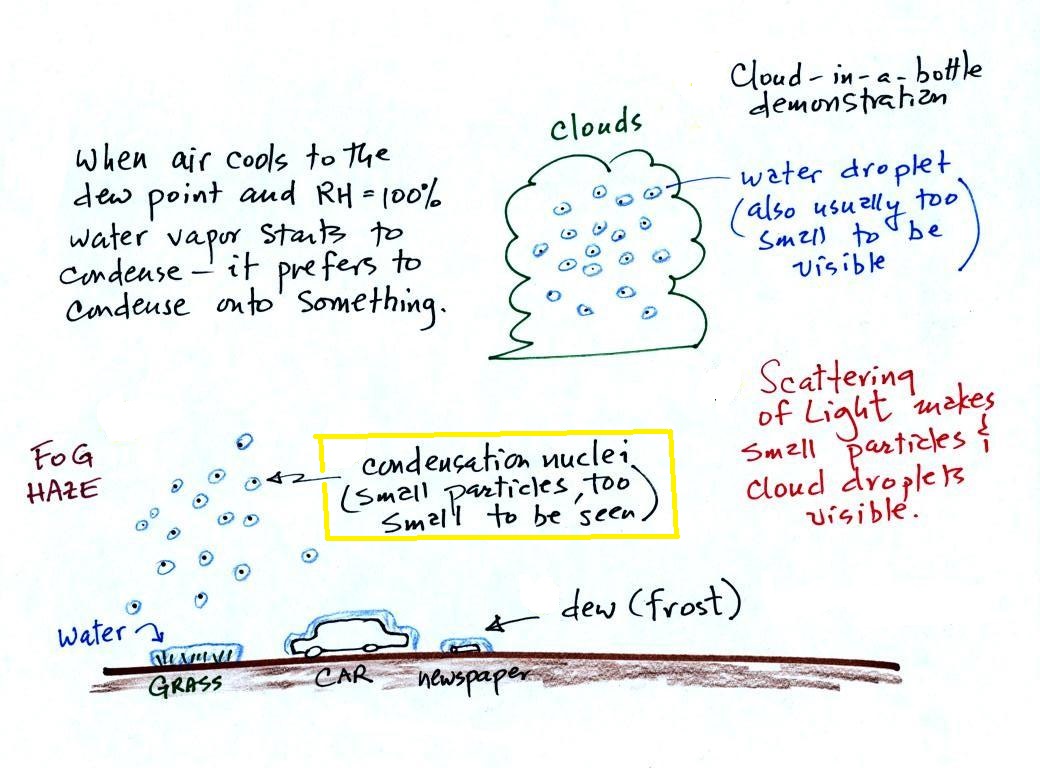
Condensation nuclei and the
formation of dew, frost, haze, fog, and clouds
Here's a visual summary of a part of what we'll be
covering next.
A variety of things can happen when you cool air to the dew
point and the relative humidity increases to 100%. When
moist air next to the ground becomes saturated (RH reaches 100%)
water vapor condenses onto (or, in the case of frost, is deposited
onto) the ground or objects on the ground. This forms dew,
frozen dew, and frost.
When air above the ground cools to the dew point, it is much
easier for water vapor to condense onto small particles in the air
called condensation nuclei. It would be much more difficult
for the water vapor to condense and form small drops of pure
water. Both the condensation nuclei and the small water
droplets that form on them are usually too small to be seen with
the naked eye. We can tell they are present because they
scatter sunlight and make the sky hazy. As humidity
increases dry haze turns to wet haze and eventually to fog.
We'll try to make a cloud in a bottle and you'll be able to better
appreciate the role that condensation nuclei play.
Condensation nuclei and the role they play in cloud
droplet formation
The air next to the ground cools during the night.
Sometimes it cools enough to reach the dew point. Water
vapor condenses onto objects on the ground and you find everything
covered with dew (or frost) the next morning. When this
happens in the air up above the ground you might think that water
vapor would simply condense and form little droplets. This
is not the case; we will find that small particles in the air
called condensation play an essential role in cloud (and fog)
formation.
it is much
easier for water vapor
to condense onto small particles
called condensation nuclei |
it would be
much harder for
water vapor
to just condense and form
small droplets of pure water
|
We probably won't go into all of the
details that follow in class, though they aren't hard to
figure out and understand. If you'd prefer to just skip
the details, just remember that particles make it easier for
cloud droplets and clouds to form.
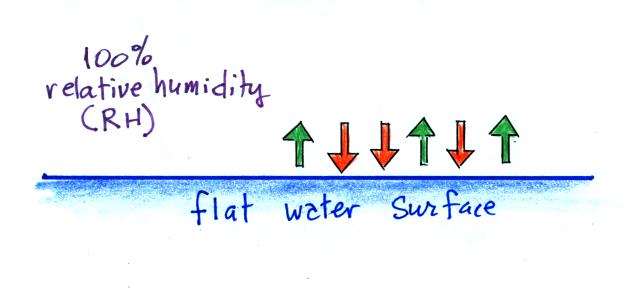
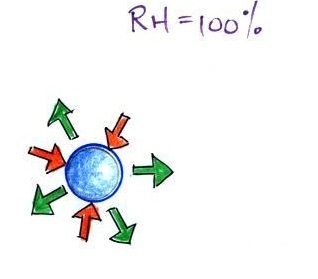
When
the air is saturated with water vapor (the
relative humidity is 100%) the rates of
evaporation and condensation above a flat
surface of water will be equal.
There's no real reason for
picking three arrows each of evaporation and
condensation, the important point is that they are
equal when the RH is 100%.
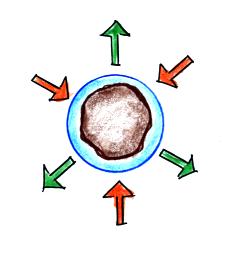
It's hard for water vapor to condense and form a
small droplet of water because small droplets
evaporate at a very high rate. This is known as
the curvature effect and is illustrated below.
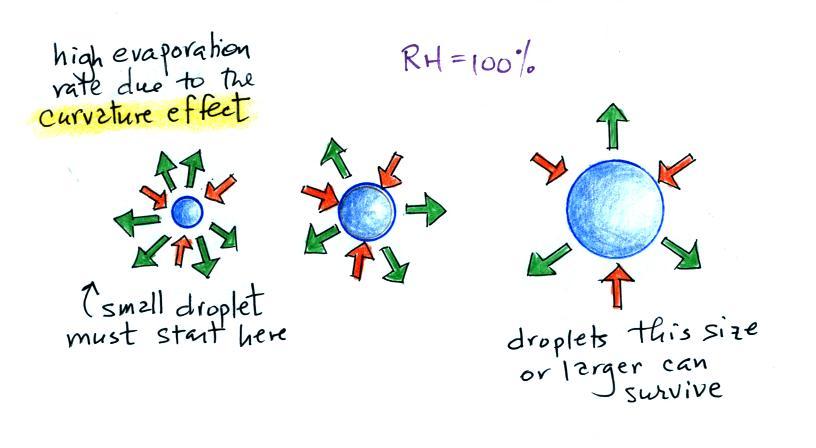
The surface of the smallest droplet above at left has
the most curvature and the highest rate of evaporation (6
arrows). If a small droplet like this were to form,
it wouldn't stay around very long. With it's high
rate of evaporation it would quickly evaporate away and
disappear.
The middle droplet is larger and would stick around a
little longer because it does not evaporate as
quickly. But it too would eventually disappear.
The drop on the right is large enough that curvature no
longer has an effect. This drop has an evaporation
rate (3 arrows) that is the same as would be found over a
flat surface of water. A droplet like this could
survive, but the question is how could it get this big
without going through the smaller sizes with their high
rates of evaporation. A droplet must
somehow reach a critical size before it will be in
equilibrium with its surroundings.
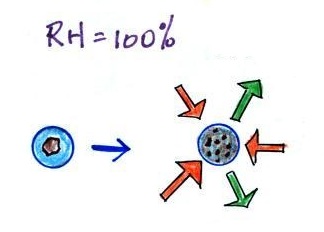
Particles in the air, cloud condensation nuclei (CCN),
make it much easier for cloud droplets to form.
The figure below explains why.
By condensing onto a particle, the water droplet starts
out large enough and with an evaporation rate low enough
that it is in equilibrium with the moist surroundings
(equal rates of condensation and evaporation).
There are always lots of CCN (cloud condensation nuclei
in the air) so this isn't an impediment to cloud
formation. |
Now back to material that we did
cover in class.
The following information is from p. 91 in the ClassNotes.

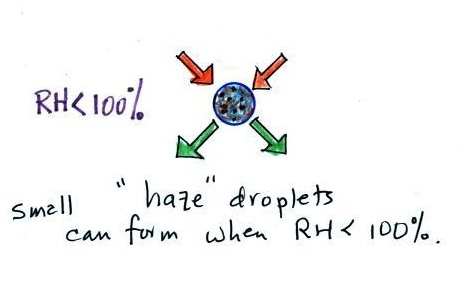
Note that condensation onto
certain kinds of condensation nuclei and growth of cloud
droplets can begin even when the relative humidity is below
100%. These are called hygroscopic nuclei. Salt
is an example; small particles of salt mostly come from
evaporating drops of ocean water.
I might try to show a video tape, not a digital video but video
recorded on a magnetic tape. It will depend first of all
on there being a VCR in the classroom.
Here are some more of the
details that we probably won't cover in class.
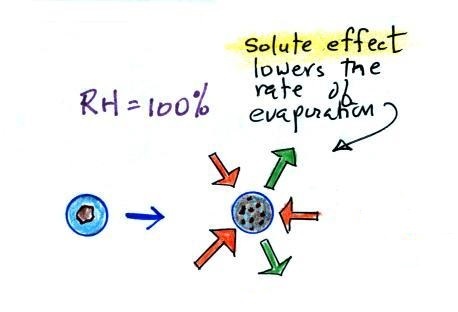
To understand how
condensation onto particles can begin even before the RH
has reached 100% we first need to learn about the solute
effect
|
|
solution
droplet
|
pure water
droplet
|
Water vapor condensing onto
the particle in the left figure dissolves the
particle. The resulting solution evaporates at a
lower rate (2 arrows of evaporation). A droplet of
pure water of about the same size would evaporate at a
higher rate (4 arrows in the figure at right).
Note the rates of condensation are equal in both figures
above. This is determined by the amount of
moisture in the air surrounding each droplet. We
assume the same moist (the RH is 100%) air surrounds
both droplets and the rates of condensation are
equal.
The next figure compares solution droplets that form
when the RH is 100% (left figure) and when the RH is
less than 100%.
|
|
the droplet is
able to grow
|
the droplet is
in equilibrium with its surroundings
even when the RH is less than 100%
|
The solution droplet will grow in the RH=100%
environment at left. You can tell the RH is less
than 100% in the figure at right because there are now
only 2 arrows of evaporation. But because the
solution droplet only has 2 arrows of evaporation it can
form and be in equilibrium in this environment.
|
We should remember that much of what we see in the sky is
caused by scattering of light. There was a pretty good
demonstration of light scattering during one of the music videos
played before class on Monday.
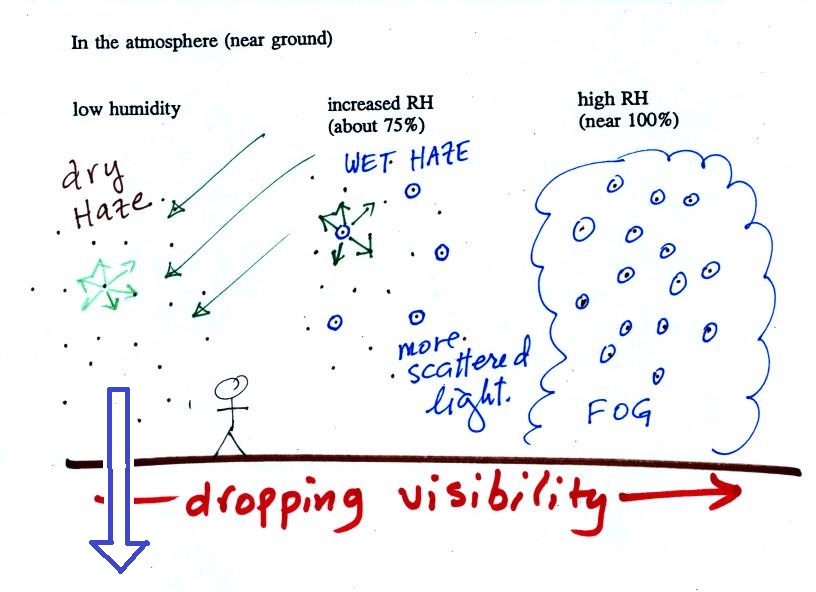
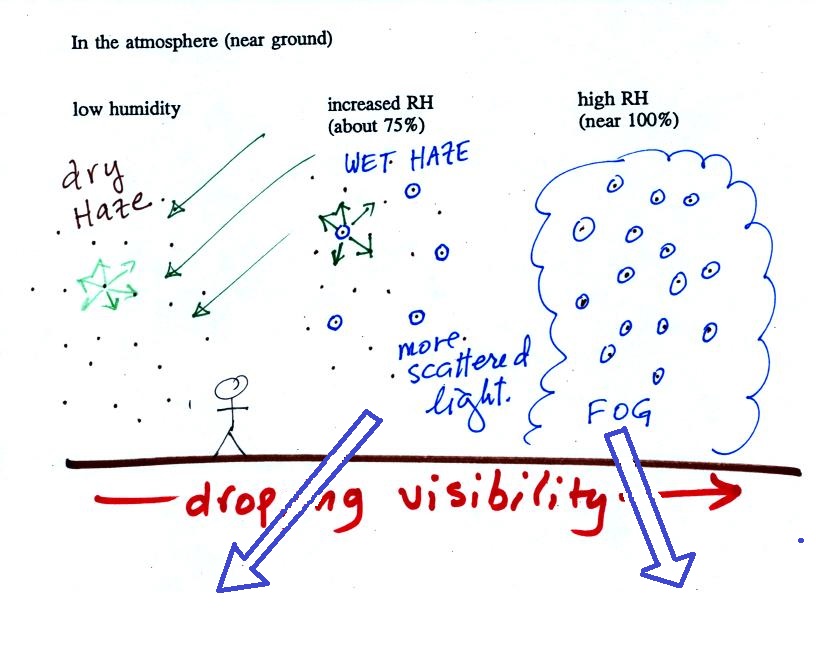
The following figure is at the bottom of p. 91 in the ClassNotes
and illustrates how cloud condensation nuclei and
increasing relative humidity can affect the appearance of the sky
and the visibility.
The air in the left most figure is relatively dry. Even
though the condensation nuclei particles are too small to be seen
with the human eye you can tell they are there because they
scatter sunlight. When you look at the sky you see the deep
blue color caused by scattering of sunlight by air molecules mixed
together with some white sunlight scattered by the condensation
nuclei. This changes the color of the sky from a deep blue
to a bluish white color. The more particles there are the
whiter the sky becomes. This is called "dry haze."
Visibility under these conditions might be anywhere from a few
miles up to a few tens of miles.
A photograph of fairly severe air pollution in
Paris that illustrates an extreme
case of dry haze (this is more common and
more severe in China and India). In Paris cars with
even numbered license plates weren't allowed into the city on
certain days of the week, odd numbers were banned on other
days. Public transportation was free for a short time to try
to reduce automobile use.
The middle picture below shows what happens when you drive from
the dry southwestern part of the US into the humid southeastern US
or the Gulf Coast. One of the first things you would notice
is the hazier appearance of the air and a decrease in
visibility. It isn't that there are more particles.
The relative humidity is higher, water vapor begins to condense
onto some of the condensation nuclei particles (the hygroscopic
nuclei) in the air and forms small water droplets. The water
droplets scatter more sunlight than just small particles
alone. The increase in the amount of scattered light is what
gives the air its hazier appearance. This is called "wet
haze." Visibility now might now only be a few miles.

|

|
Thin fog
(perhaps even wet haze)
with pretty good visibility
(source
of the image)
|
Thick fog
(visibility was less than 500 feet)
(source
of the image)
|

|
 |
Pictures of fog
like we sometimes get in Tucson (maybe once a year).
The picture at left is looking east from my house and was
taken early in the morning at the start of the spring
semester in 2015. The picture at right is the view
to the west. Visibility was perhaps 1/4 mile.
|
Finally when the relative humidity increases to 100% fog forms
and water vapor condenses onto all the condensation nuclei.
Fog can cause a severe drop in the visibility. The thickest
fog forms in dirty air that contains lots of condensation
nuclei. That is part of the reason the Great London Smog of
1952 was so impressive. Visibility was at times just a few
feet!
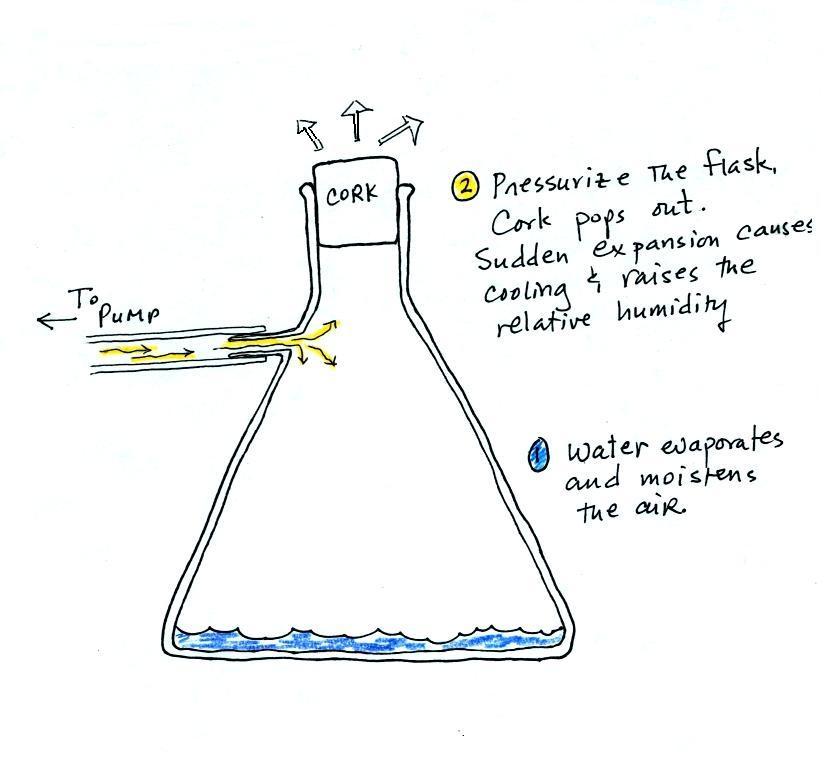
Making a cloud in a bottle
Cooling air & increasing relative humidity, condensation
nuclei, and scattering of light
are all involved in this demonstration.
We used a
strong, thick-walled, 4 liter vacuum flask (designed
to not implode when all of the air is pumped out of
them, they really aren't designed to be
pressurized). There was a little water in the
bottom of the flask to moisten the air in the
flask. Next we pressurized the air in the
flask with a bicycle pump. At some point the
pressure blows the cork out of the top of the
flask. The air in the flask expands outward
and cools. This sudden cooling increases the
relative humidity of the moist air in the flask to
more than 100% momentarily and water vapor condenses
onto cloud condensation nuclei in the air.
I like it best when a faint, hard to see, cloud
becomes visible. That's because there is
something we can add to the demonstration that will
make the cloud much "thicker" and easier to see.
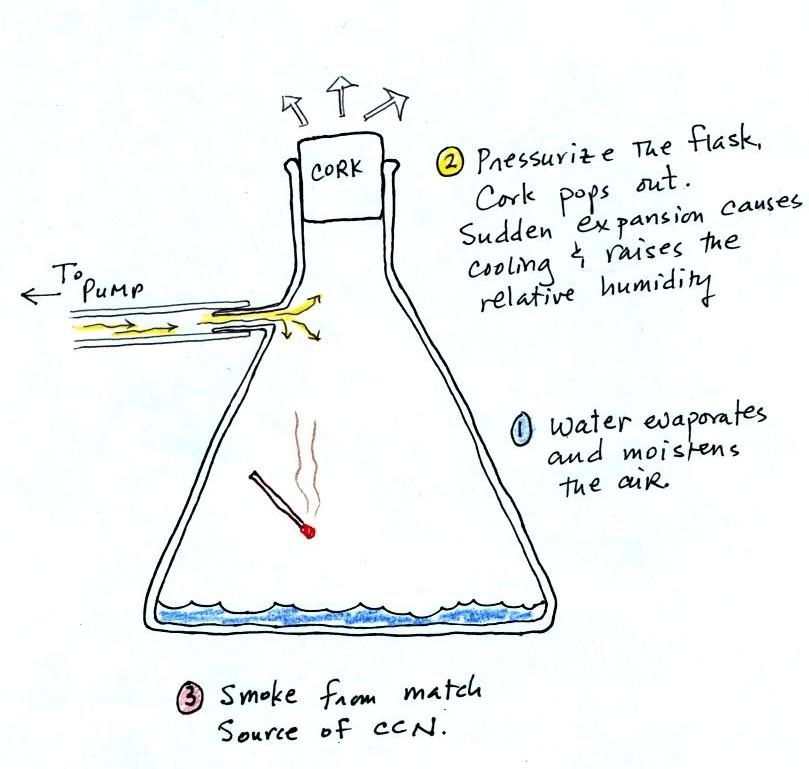
The demonstration was repeated an additional
time with one small change. A burning match was dropped
into the bottle. The smoke from the matches added lots of
very small particles, condensation nuclei, to the air in the
flask (you could see the swirls of smoke, the small particles
scattered light). The same amount of water vapor was
available for cloud formation but the cloud that formed this
time was quite a bit "thicker" and much easier to see. To
be honest the burning match probably also added a little water
vapor (water vapor together with carbon dioxide is one of the by
products of combustion).
I have found a couple of online versions of the
demonstration. The first
is performed by Bill Nye "The Science Guy" and is pretty similar
to the one done in class. The second
differs only in the way that is used to caused the sudden
expansion and cooling of the air (I didn't care much for the
music (probably your opinion of the music I play before class)
and I would recommend turning down the sound while watching the
video).
Mother Nature's version of the Cloud in a Bottle
demonstration
A
brush fire in this picture is heating up air and causing
it to rise. Combustion also adds some moisture and
lots of smoke particles to the air. You can see
that initially the rising air doesn't form a cloud (the
RH is still less than 100%). A little higher and
once the rising air has cooled enough (to the dew point)
a cloud does form. And notice the cloud's
appearance - puffy and not a layer cloud. Cumulo
or cumulus is the word used to describe a cloud with
this appearance. These kinds of fire caused clouds
are called pyrocumulus clouds. The example above
is from a
Wikipedia article fire-caused clouds.
The fire in this case was
the "Station Fire" burning near Los Angeles in August
2009. We sometimes see clouds like this in the summer
when lightning starts a fire burning in one of the nearby
forests. The pyrocumulus cloud caused by the fire is
sometimes the only cloud in the sky.
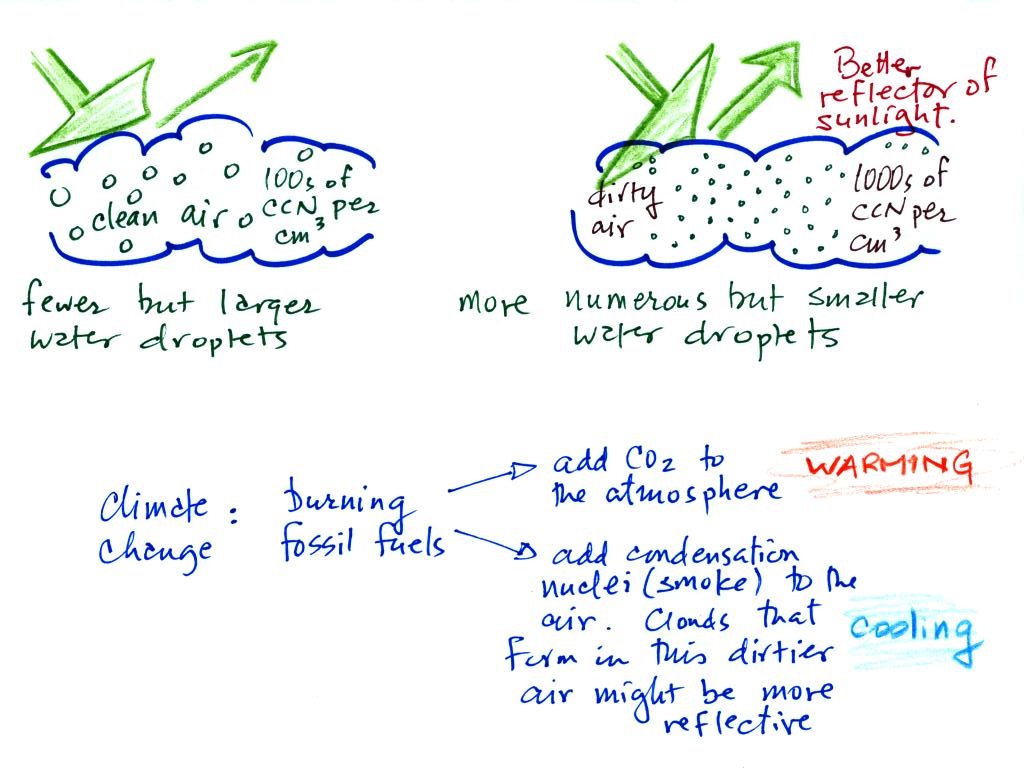
Clouds and climate change
This effect has some implications for climate change.
A cloud that forms in dirty
air is composed of a large number of small droplets (right
figure above). This cloud is more reflective than a cloud
that forms in clean air, that is composed of a smaller number of
larger droplets (left figure).
Combustion of fossil fuels adds carbon dioxide to the
atmosphere. There is concern that increasing carbon
dioxide concentrations (and other greenhouse gases) will enhance
the greenhouse effect and cause global warming. Combustion
also adds condensation nuclei to the atmosphere (just like the
burning match added smoke to the air in the flask). More
condensation nuclei might make it easier for clouds to form,
might make the clouds more reflective, and might cause
cooling. There is still quite a bit of uncertainty about
how clouds might change and how this might affect climate.
Remember that clouds are good absorbers of IR radiation and also
emit IR radiation.
Clouds are one of the best ways of cleaning the
atmosphere. This is something we mentioned earlier in the
semester and you're now in a position to understand it better.
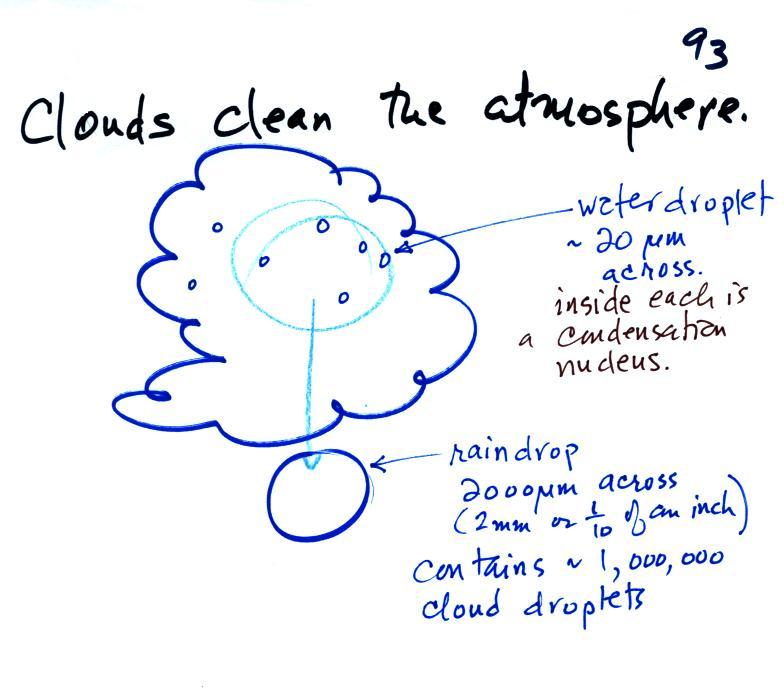
A cloud is composed of small water droplets (diameters of 10 or
20 micrometers) that form on particles ( diameters of perhaps
0.1 or 0.2 micrometers). The droplets "clump" together to form a
raindrop (diameters of 1000 or 2000 micrometers which is 1 or 2
millimeters), and the raindrop carries the particles to the
ground. A typical raindrop can contain 1 million cloud
droplets so a single raindrop can remove a lot of particles from
the air. You may have noticed how clear the air seems the
day after a rainstorm; distant mountains are crystal clear and
the sky has a deep blue color. Gaseous pollutants can
dissolve in the water droplets and be carried to the ground by
rainfall also. We'll be looking at the formation of
precipitation in more detail later this week.
We'll see how we're doing on time at
this point.
If there's sufficient time we'll go over to the Mon., Mar. 26 notes and get started
on Cloud Identification and Classification