The recycling of elements among the various components of the Earth system is key to the continued functioning of the Earth as a living planet. Although many elements are critical, none is more central to the workings of the Earth system than carbon. All life (at least on Earth) is based on carbon. In fact carbon makes up about 50% of our dry weight (excluding water). Ecologists study ecosystems by tracking carbon exchanges through the system. Both biological and physical processes are involved in the recycling of carbon, and they are so closely intertwined that it is difficult to separate the two. In this class we will do no more than look at a simplified overview of the carbon cycle focussing mainly on the atmospheric reservoir of carbon.
Beside being the central constituent of all organic compounds, carbon in the atmosphere, which is mostly in the form carbon dioxide, is an important greenhouse gas. Without the greenhouse effects of carbon dioxide in the atmosphere, life as we know it would not exist on Earth because the surface temperatures would be too cold. However, the actions of man have disrupted the natural carbon cycling by causing imbalances in the flow of carbon to and from the atmospheric reservoir. The net effect has been a sharp increase in the amount of carbon dioxide stored in the atmosphere. The biggest concern with this is how will the extra carbon dioxide affect the Earth's climate and in turn its life? As we will see this is a very difficult question to answer due to the complexities of both the carbon cycle and the climate system, which are interlinked. The ultimate outcomes stemming from anthropogenic perturbations to the carbon cycle remain quite uncertain. Thus, what, if anything, should be done about the human-caused increases in the carbon dioxide concentrations in the atmosphere remains a hotly debated issue.
In lecture, we will draw simplified budget diagrams for carbon. The first diagram will represent the carbon budget before the industrial revolution. Later the anthropogenic perturbations due to fossil fuel burning and deforestation will be added. The paragraphs that follow describe some of the reservoirs and fluxes involved in the carbon budget.
Carbon dioxide (CO2), a colorless gas having a faint, sharp odor and a sour taste; it is a minor component of the Earth's atmosphere (presently about 379 parts per million, which is up from about 280 ppm at the time of the industrial revolution).
The source of the carbon found in living matter is carbon dioxide (CO2) in the air or dissolved in water.
Algae and terrestrial green plants (producers) are the chief agents of carbon dioxide fixation through the process of photosynthesis, in which carbon dioxide and water are converted into simple carbohydrates.
Carbohydrates are used by the producers to carry on metabolism, the excess being stored as fats and polysaccharides. The stored products are then eaten by consumer animals, from protozoan to man, which convert them into other forms.
All animals return CO2 directly to the atmosphere as a by-product of their respiration.
The carbon present in animal wastes and in the bodies of all organisms is released as CO2 by decay, or decomposer organisms (chiefly bacteria and fungi) in a series of microbial transformations.
Part of the organic carbon--the remains of organisms--has accumulated in the Earth's crust as fossil fuels (e.g., coal, gas, and petroleum), limestone, and coral. The flux linkages between the atmosphere and lithosphere were probably closely balanced before the industrial revolution. Note how slow these processes are (small fluxes) compared withe exchanges of carbon between the atmosphere and oceans or land.
Carbon dioxide is continuously exchanged between the atmosphere and the ocean. The distribution of sources and sinks of CO2 is tied to the circulation and productivity patterns of the oceans. In regions of the ocean where there are high rates of primary productivity (building of organic material through photosynthesis), there is a net flux of carbon from the atmosphere into the oceans. Conversely, in upwelling regions where the ocean water is CO2 rich, the net flux of carbon is from the oceans into the atmosphere. Before the carbon cycle was disturbed by human activities, the flux of CO2 into the ocean was probably closely balanced by the flux out.
After 1850, humans began to significantly alter the balanced carbon cycle through burning fossil fuels and deforestation. The carbon stored in fossil fuels is now being released in vast amounts as CO2 when these fossil fuels are "burned" or used as energy sources. Deforestation adds carbon dioxide to the atmosphere through two processes: (1)Trees that have been removed, no longer participate in photosynthesis which removes CO2 from the atmosphere and (2)When these trees are burned or decay, they release all the stored carbon they contain. In lecture, these processes will be added to the pre-industrial carbon budget diagram.
We can fairly well compute the rate of carbon release to the atmosphere by fossil fuel burning and deforestation. And if these were the only changes to the carbon budget, we could easily calculate how fast the atmospheric reservoir of carbon would increase. However, the system itself (and any intertwined systems) responds to these human perturbations. We do not understand the details and complexities of the carbon cycle well enough to accurately predict how the various fluxes and reservoirs will change with time.
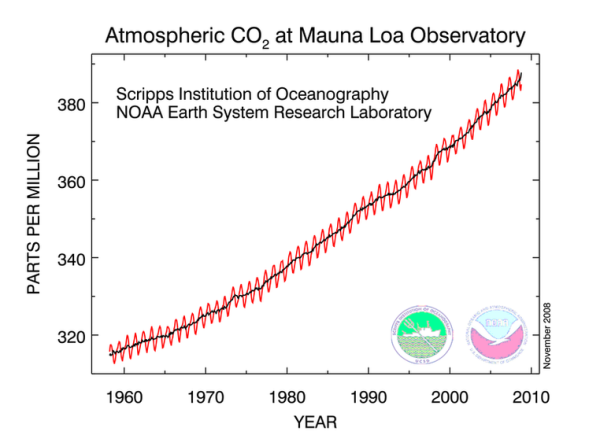
One thing that we do know for sure is that the concentration of CO2 in the atmosphere is increasing. We have very accurate measurements taken at Mauna Loa, HI since the late 1950s. Prior to that we can estimate the atmospheric CO2 concentrations from ice core samples.
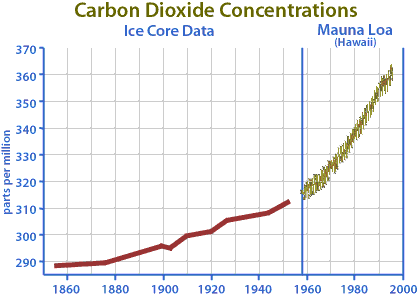
|
|
| Close-up Atmospheric CO2 concentration from Mauna Loa |
No doubt that CO2 levels in the atmosphere are increasing. The year to year fluctuations are tied to the seasonal cycle of plant growth in the Northern Hemisphere's terrestrial ecosystems. In the Northern hemisphere summer, land plants grow by photosynthesis, taking in lots CO2, and reduce the CO2 in the atmosphere. Conversely, in the Northern hemisphere winter, photosynthetic activity decreases, but the processes of respiration and decay continue, thus CO2 levels in the atmosphere increase.
Scientists have developed models of the carbon cycle to make predictions of how the system will respond to the human perturbations. One response that we are reasonably sure about is that the oceanic uptake of CO2 has increased in response to the increase in the atmospheric concentrations of CO2. However, there remains a problem: Our models of the carbon cycle predict that the present day concentration CO2 in the atmosphere should be 467 parts per million, but the measured amount is 390 ppm; therefore, more CO2 is being removed from the atmosphere than is predicted. This discrepancy has been called the "missing sink" problem for atmospheric CO2.
For the decade of the 1990s, the global carbon cycle can be summarized as follows (units are PgC. One Pg [petagram] = one billion metric tonnes = 1000 x one billion kg):
| Atmospheric increase | = | Emissions from Fossil fuels | + | Net emissions from changes in land use | - | Oceanic uptake | - | "Missing" carbon sink |
| 3.2 (±0.2) | = | 6.3 (±0.4) | + | 2.2 (±0.8) | - | 2.4 (±0.7) | - | 2.9 (±1.1) |
Attention on the global carbon cycle over more than 25 years has focused on the apparent imbalance in the carbon budget in the above equation -- the so-called "missing sink," missing because the accumulation of carbon has not been observed. Breaking down the numbers given above we can say: Of all the CO2 added to the atmosphere by human activity, roughly 40% remains in the atmosphere, 30% is absorbed by the oceans, and the remaining 30% is the "missing sink". Perhaps surprisingly, those percentages seem to remain roughly the same, regardless of fluctuations in the amount of CO2 that is added to the atmosphere by human activities.
The problem of the missing CO2 sink has still not been satisfactorily explained. Where is this "missing" carbon going? The most accepted explanation, though not scientifically proven, is that much of the missing carbon is being taken up and stored in land plants. It appears that photosynthesis has been faster than decomposition for decades although we are still unable to say for sure exactly where the carbon is stored. There are several possible contributing factors. One is that plants may be growing faster due to CO2 fertilization. Another possibility is related to global warming. Global average temperatures have increased (especially since the 1980s). This seems to have extended the growing season in northern forests. The longer growing season could allow trees to take in more CO2. Regrowth of natural forests after agricultural abandonment, particularly assoicated with small family farms, may be removing more CO2 than thought. Some also point to suppression of forest fires as a possible contributing factor in keeping CO2 out of the atmosphere. These explanations seem reasonable considering that forests contain about 90% of all of the living biomass on land. Those of you who have followed the global warming debate closely, know that part of the Kyoto Treaty contains "carbon credits" for countries that plant new trees that should effectively remove CO2 from the atmosphere.
Finally, keep this in mind as we discuss the global warming issue: One of the many uncertainties in the prediction of future global warming is our poor understanding of the carbon cycle itself. Even though we know how much CO2 is being added to the atmosphere by fossil fuel burning, we are not able to predict with certainty what happens to all of it. Of course, this limits our ability to predict how much warming may occur.
The Random House Dictionary defines a pollutant as "any substance, as certain chemicals or waste products, that renders the air, soil, water, or other natural resource harmful or unsuitable for a specific purpose." In 2007, the United States Supreme Court Decided that CO2 is a pollutant. However, since CO2 is not directly harmful to breathe, even at concentrations over 100 times greater than the current atmospheric concentrations (which incidentally is the approximate CO2 concentration in our exhaled breath), the only way the Supreme Court can make that ruling is to say that the potential climate changes that may come about due to CO2 will render the Earth harmful or unsuitable for people and other life. In my mind, we do not understand the Earth's climate system well enough to make that prediction. The court's ruling probably has more to do with giving government authority to regulate CO2 emissions. You may well agree with that, but it is plain wrong to classify CO2 as a pollutant in the same category as a truly toxic gas like carbon monoxide. In fact CO2 is absolutely essential for plants and as a result for most life on this planet to exist.
Some sanity is brought to this issue in Princeton University Physicist Dr. Will Happer's Statement before the US Senate's Environment and Public Works Full Committee Hearing entitled "Update on the Latest Global Warming Science." A paragraph from that testamony is quoted below.
"I keep hearing about the pollutant CO2, or about poisoning the atmosphere with CO2, or about minimizing our carbon footprint. This brings to mind another Orwellian pronouncement that is worth pondering: But if thought corrupts language, language can also corrupt thought. CO2 is not a pollutant and it is not a poison and we should not corrupt the English language by depriving pollutant and poison of their original meaning. Our exhaled breath contains about 4% CO2. That is 40,000 parts per million, or about 100 times the current atmospheric concentration. CO2 is absolutely essential for life on earth. Commercial greenhouse operators often use CO2 as a fertilizer to improve the health and growth rate of their plants. Plants, and our own primate ancestors evolved when the levels of atmospheric CO2 were about 1000 ppm, a level that we will probably not reach by burning fossil fuels, and far above our current level of about 389 ppm. We try to keep CO2 levels in our U.S. Navy submarines no higher than 8,000 parts per million, about 20 times the current atmospheric levels. Few adverse effects are observed at even higher levels."