
We first need to finish up an example that we didn't have time
for in an earlier class. It deals with what happens
along an air-cloud boundary when there is an abrupt change in
conductivity.

Conductivity inside a cloud is lower than in the air outside a
cloud. This is because the small ions attach to much
larger and much less mobile cloud particles (water droplets or
ice crystals). The E field must become stronger inside
the cloud so that the current density (the produce of
conductivity and electric field) stays the same inside and
outside the cloud. We'll see that layers of charge build
up on the top and bottom surfaces of the cloud.
In
the figure above, Z2 is just above and Z1
just below the upper edge of the cloud. We'll try to
estimate how much charge is necessary along the upper edge of
the cloud itself. We start with the continuity equation
and assume steady state conditions. Both λ and E
are functions of z only.

We
don't really know Ez. The current
density, Jz, on
the other hand is constant with altitude and we assume we know
how conductivity changes as you move across the cloud-air
boundary.
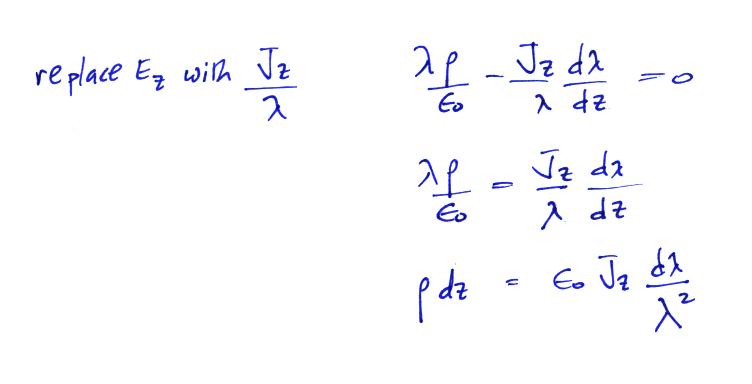
We
can integrate this equation
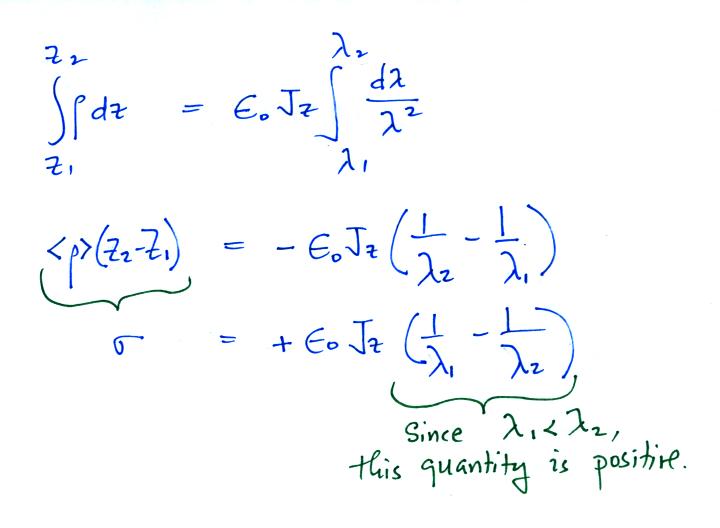
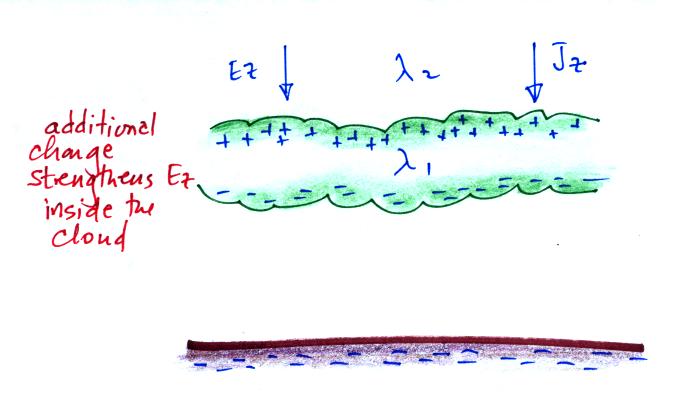
The effect of these two layers of cloud is to intensify the
field inside the cloud. The product of higher field
times lower conductivity inside the cloud is able to keep the
current density equal to the current density outside the
cloud.
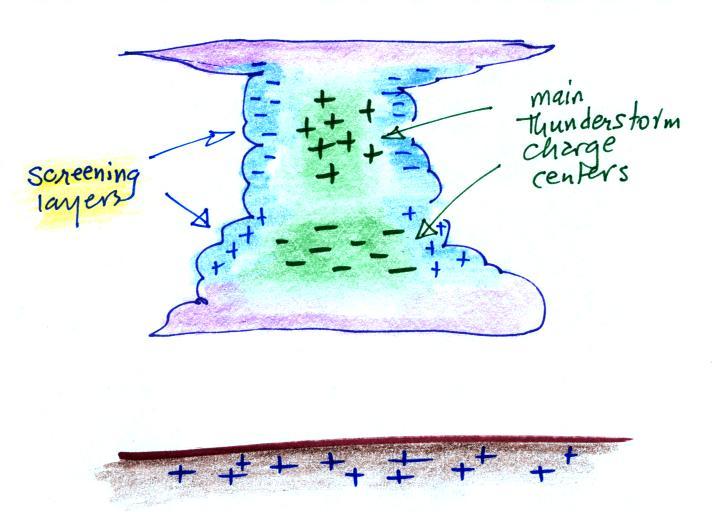
Screening layers that form along the edges of a thunderstorm
effectively mask the main charge centers inside the cloud.

The free electrons attach to oxygen molecules in a few to a few tens of nanoseconds (1 ns = 10-9 seconds). Click here if you'd like to read more about how this "attachment time" is determined.
Then water vapor molecules cluster around the ions to create
"small ions." Water molecules have a dipole structure as
shown below.
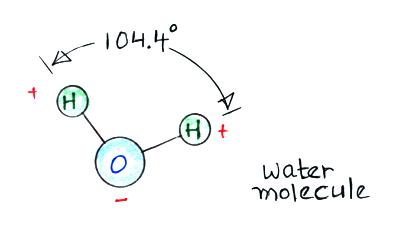
The oxygen atom carries excess negative charge and the
hydrogen atoms positive charge. Because of this the
water vapor molecules orient themselves differently around the
oxygen and nitrogen ions. This takes a few milliseconds
to occur (we won't look at how that is determined).
Conceptually this would look like

More
water
vapor molecules are able to surround the positive ions so they
are bigger and have slightly lower electrical mobility than
the negative small ions (typical values are included so that
you can get some feel for the difference).
Sources
of atmospheric ionization
The next figure summarizes the processes that ionize
air. This is new information.
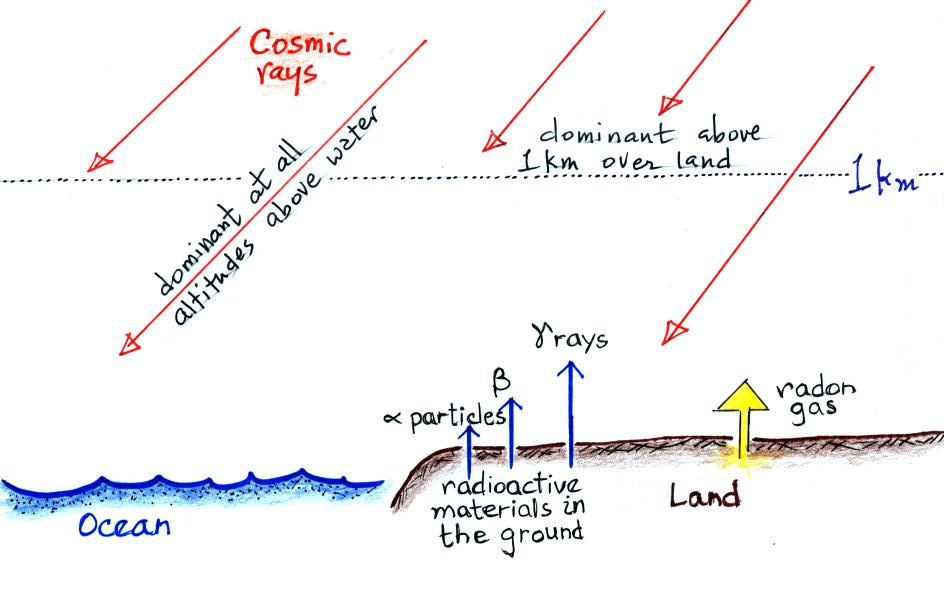
Radioactive materials in the ground emit alpha and beta
particles, and gamma rays. Alpha particles (i.e. a
helium nucleus consisting two protons and two neutrons) are a
strong source of ionization but only in the first few cm above
the ground. Beta particles (electrons) ionize air in a
layer a few meters thick. The effects of gamma radiation
extend of 100s of meters. Cosmic rays are the dominant
source of ionization everywhere over the ocean and at greater
than 1 km altitude over land.
The table below gives an idea of how far these different types
of radiation can travel above the ground and also typical
ionization rates (ip stands for
"ion pairs"). (from Chapter 11 in "The Earth's Electrical
Environment," National Academy of Sciences, 1986 )
|
emission type |
distance of travel |
ionization rate [ ip/(cm3
sec) ] |
|
alpha particles |
only a few cm above the ground |
not well known |
|
beta particles |
a few meters above the ground |
0.1 to 10 |
|
gamma rays |
100s of meters above the ground |
1 to 6 |
|
radon |
depends on atmospheric conditions |
1 to 20 at 1-2 m above ground |
|
cosmic rays |
1 to 2 ip/(cm3
sec) near the ground |
|
Cosmic
radiation
We'll
start with some information about cosmic radiation (cosmic
rays). This is the dominant ionization process over
the oceans and over land at altitudes above 1 km.




Cosmic-ray
air showers
Cosmic
rays
The
earth is hit by elementary particles and
atomic nuclei of very large energies.
Most of them are protons (hydrogen
nuclei) and all sorts of nuclei up to
uranium (although anything heavier than
nickel is very, very rare). Those are
usually meant when talking about cosmic
rays. Other energetic particles in the
cosmos are mainly electrons and
positrons, as well as gamma-rays and
neutrinos.
Interactions
and particle production
The
cosmic rays will hardly ever hit the
ground but will collide (interact) with
a nucleus of the air, usually several
ten kilometers high. In such collisions,
many new particles are usually created
and the colliding nuclei evaporate to a
large extent. Most of the new particles
are pi-mesons (pions). Neutral pions
very quickly decay, usually into two
gamma-rays. Charged pions also decay but
after a longer time. Therefore, some of
the pions may collide with yet another
nucleus of the air before decaying,
which would be into a muon and a
neutrino. The fragments of the incoming
nucleus also interact again, also
producing new particles.
The gamma-rays
from the neutral pions may also create new
particles, an electron and a positron, by
the pair-creation process. Electrons and
positrons in turn may produce more
gamma-rays by the bremsstrahlung
mechanism.
Shower
development
The number
of particles starts to increase rapidly as
this shower or cascade of particles moves
downwards in the atmosphere. On their way
and in each interaction the particles
loose energy, however, and eventually will
not be able to create new particles. After
some point, the shower maximum, more
particles are stopped than created and the
number of shower particles declines. Only
a small fraction of the particles usually
comes down to the ground. How many
actually come down depends on the energy
and type of the incident cosmic ray and
the ground altitude (sea or mountain
level). Actual numbers are subject to
large fluctuations.
In fact, from
most cosmic rays nothing comes down at
all. Because the earth is hit by so many
cosmic rays, an area of the size of a hand
is still hit by about one particle per
second. These secondary cosmic rays
constitute about one third of the natural
radioactivity.
When a primary
cosmic ray produces many secondary
particles, we call this an air shower.
When many thousand (sometimes millions or
even billions) of particles arrive at
ground level, perhaps on a mountain, this
is called an extensive air shower (EAS).
Most of these particles will arrive within
some hundred meters from the axis of
motion of the original particle, now the
shower axis. But some particles can be
found even kilometers away. Along the
axis, most particles can be found in a
kind of disk only a few meters thick and
moving almost at the speed of light. This
disk is slightly bent, with particles far
from the axis coming later. The spread or
thickness of the disk also increases with
distance from the axis.
Shower detection
Extensive
air showers with many particles arriving on
the ground can be detected
with different kinds of particle detectors.
In the air the particles may also emit light
by two different processes: Cherenkov light
almost along the shower axis and
fluorescence light in all directions.
Cosmic Rays
by Richard Mewaldt
Cosmic Rays
by James Schombert
Further reading found on the net
(Postscript or PDF format):
Particle Data Group:
review of cosmic rays
Introduction to high
energy cosmic ray physics
Cosmic Ray Spectrum
and Composition: Ground Observations
This
page was written by Konrad Bernlöhr.
Radioactive
decay and radon
In addition to being a source of atmospheric ionization, radon
is a significant health hazard
and is the 2nd leading cause of lung cancer after
cigarettes. Here are links to articles concerning radon
from the World Health Organization,
Wikipedia, and the Environmental Protection Agency.
The following table shows a portion of the decay series that
ultimately yield isotopes of radon (the half life is shown in
parenthesis). The elements in the final row are stable.
|
|
|
|
|
|
|
|
|
|
|
|
|
|
|
Because
of its relatively short half life compared to the age of the
earth, all the Neptunium in the ground has decayed away.
Two isotopes of radon (Rn-222 and Rn-220, indicated with *)
have half lives long enough to be able to diffuse out of the
soil and into the air.
The article from the World Health
Organization gives a typical outdoor radon
concentration of 5 to 15 Becquerels/m3
(Bq/m3). 1
Becquerel is one disintegration per second. This is
something you could measure with a detector of some kind,
maybe a Geiger counter. This is not really a
concentration, rather a decay rate (dN/dt in the equation
below). We can do a calculation to see what this implies
in terms of radon concentration and ion pair production rate.
The rate at which a radioactive material decays is described
by the following equation

(note: so far in this
course we have used λ to represent linear charge
density, atmospheric conductivity, and now decay
constant).
We can solve the equation above to give

It
is easy to relate the half life, t1/2, and
the decay constant λ

The
Rn-222
isotope has a half-life of 3.8 days.

Now that we know the decay constant we'll substitute back into
the decay rate equation to determine the radon concentration
needed to produce an average outdoors decay rate of 10 Bq/m3.

(the number density for air, 2.67 x 1019
air molecules/cm3 is sometimes known as Loschmidt's number).

We can divide these two numbers to determine the number of ion
pairs produced by each distintegration.
Then we multiply by the Rn
concentration and the decay constant (which give the decay
rate) to determine the ionization rate.


These decay products can attach to dust particles which are
then inhaled and stay trapped in the lungs. Since the
decay products are themselves radioactive, long term exposure
can ultimately lead to lung cancer. Radon is apparently
the 2nd leading cause of lung cancer in the US after cigarette
smoking.
Radon concentration indoors can build to levels that are much
higher than normally found outdoors. An extreme case is
mentioned below.
