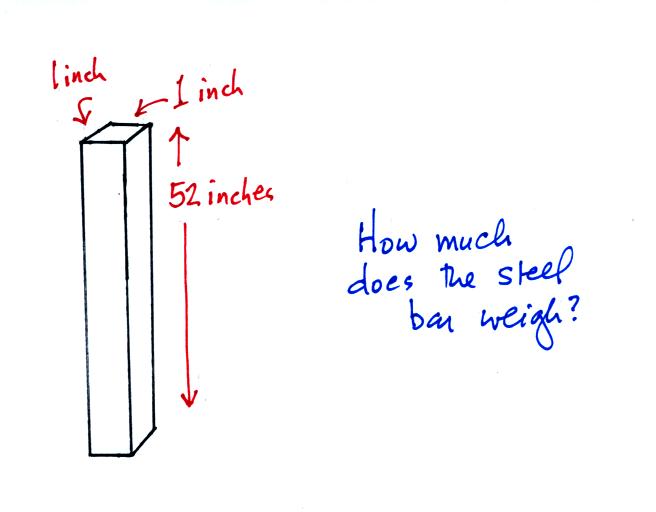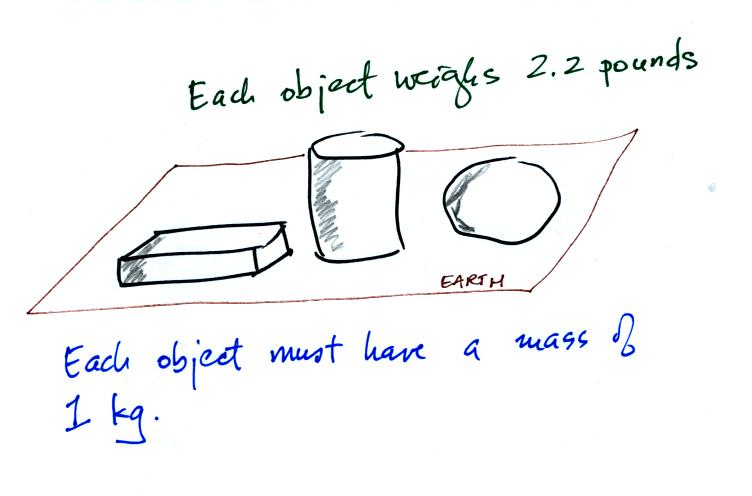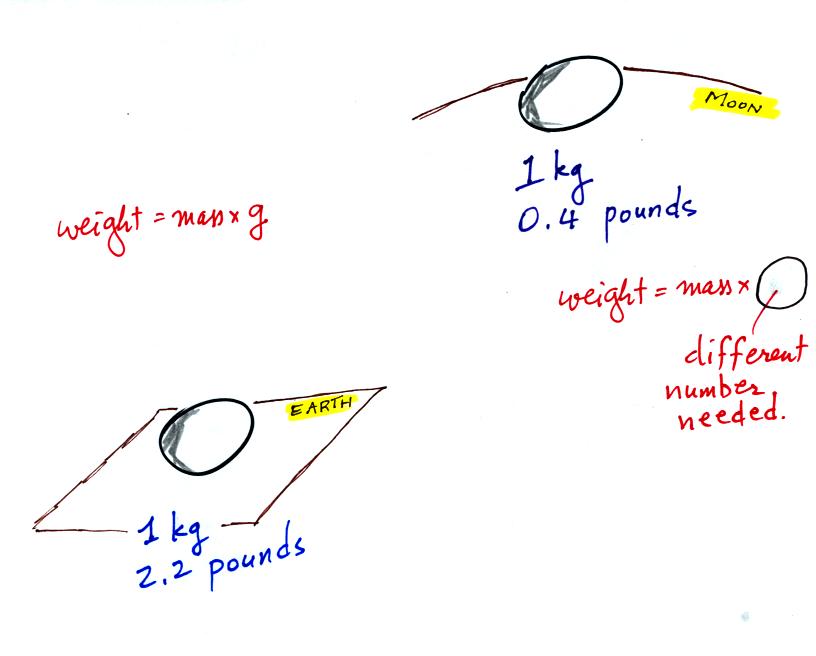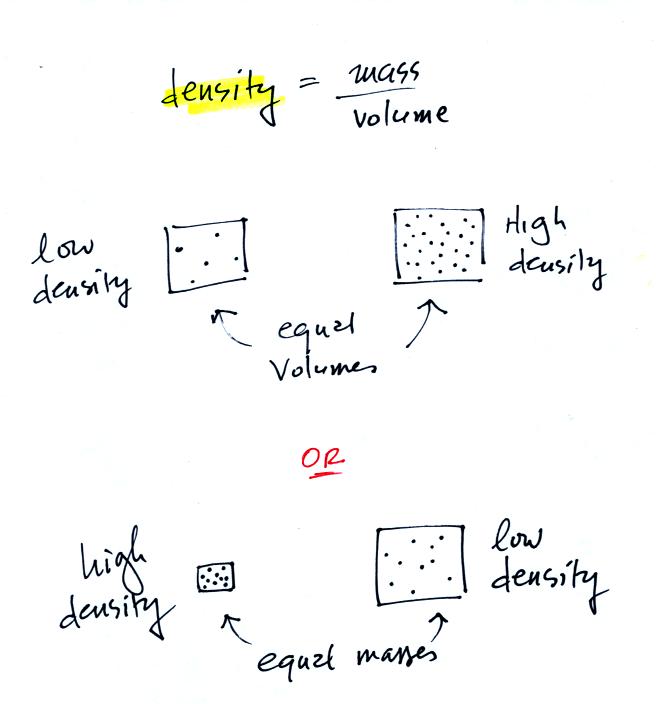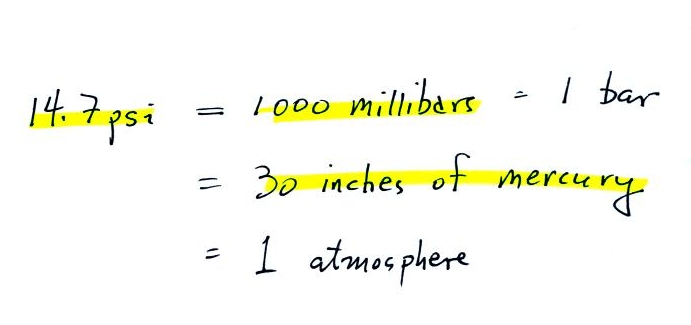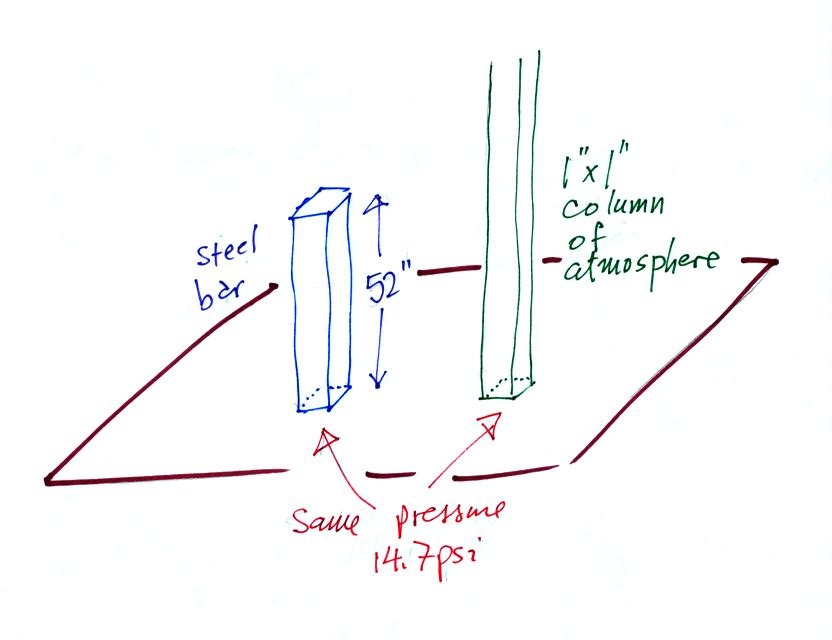
Now here's where the steel bar
comes in. The steel bar also weighs exactly 14.7 pounds (many
people thought it was heavier than that). Steel is a lot denser
than air, so a steel bar only needs to be
52 inches tall to have the same weight as an air column that is 100
miles or more tall.
Here are some of the other commonly used pressure
units.
Typical sea level
pressure is 14.7 psi or about 1000 millibars
(the
units used by meterologists and the units that we will use in this
class most of the time) or about 30 inches of mercury (refers to
the reading on a mercury barometer). If you ever find
yourself in France needing to fill your
automobile tires with air (I lived in France for a while and owned
a Peugeot
404)
remember that the air compressor scale is
probably calibrated in bars. 2 bars of pressure would be
equivalent to 30 psi.
The word "bar" basically means
pressure and is used in a lot of meteorological terms (the figure above wasn't shown in class).
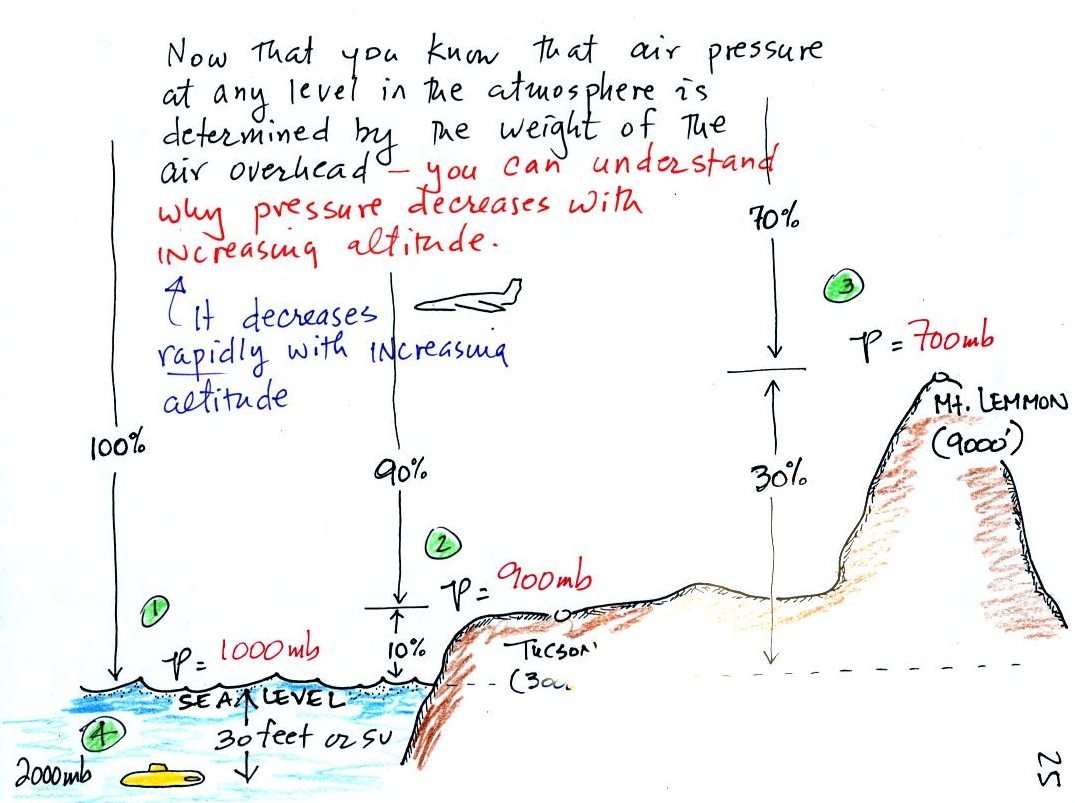
Pressure
at sea level is determined by the weight of the air overhead.
What about pressure at some level above sea level?
We can use a stack of bricks to try to
answer this
question.
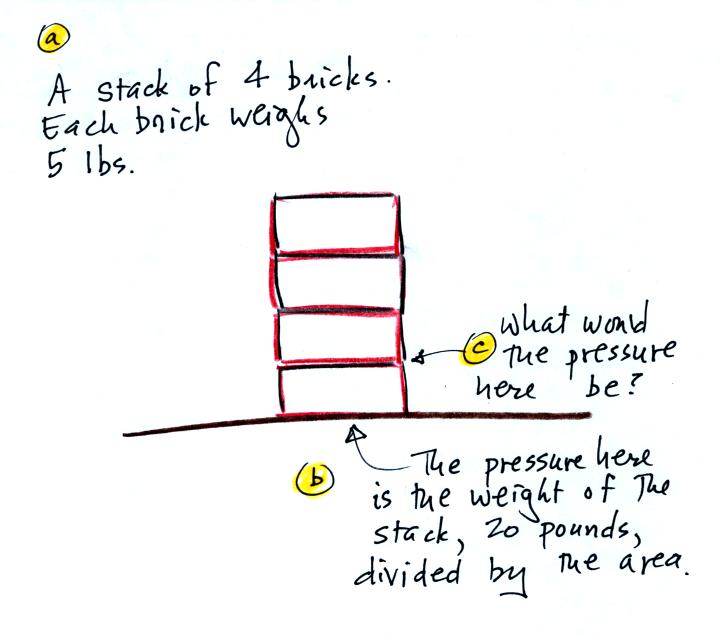
Each brick
weighs 5 pounds. At the bottom of the 4 brick tall pile you would
measure a weight of 20 pounds. If you moved up a brick you would
measure a weight of 15 pounds, the weight of the three bricks still
above. To get the pressure you would need to divide by the
area. It should be clear that weight and pressure will decrease
as you move up the pile.
In the atmosphere, pressure at any level is determined by
the weight of the air still overhead. Pressure decreases with
increasing altitude because there is less and less air remaining
overhead. The numbered points on the figure below were added
after class.
At sea
level altitude, at Point 1,
the pressure is normally about 1000 mb. That is determined by the
weight of all (100%) of the air in the atmosphere.
Some parts of Tucson, at Point 2, are 3000
feet above sea level (most
of the valley is a little lower than that). At 3000 ft. about 10%
of the
air is
below, 90% is still overhead. It is the weight of the 90% that is
still above that determines the atmospheric pressure in Tucson.
If 100% of the atmosphere produces a pressure of 1000 mb, then 90% will
produce a pressure of 900 mb.
Pressure is typically about 700 mb at the
summit of Mt. Lemmon (9000
ft. altitude at Point 3) and 70% of the atmosphere is overhead..
Pressure decreases rapidly with increasing
altitude. We will find that pressure changes more slowly if you
move horizontally. It is small horizontal changes that cause the
wind to blow however.
Point 4 shows a submarine at a depth of
about 33 ft. The pressure
there is determined by the weight of the air and the weight of the
water overhead. Water is much denser and much heavier than
air. At 33 ft., the pressure is already twice what it would be at
the surface of the ocean (2000 mb instead of 1000 mb).
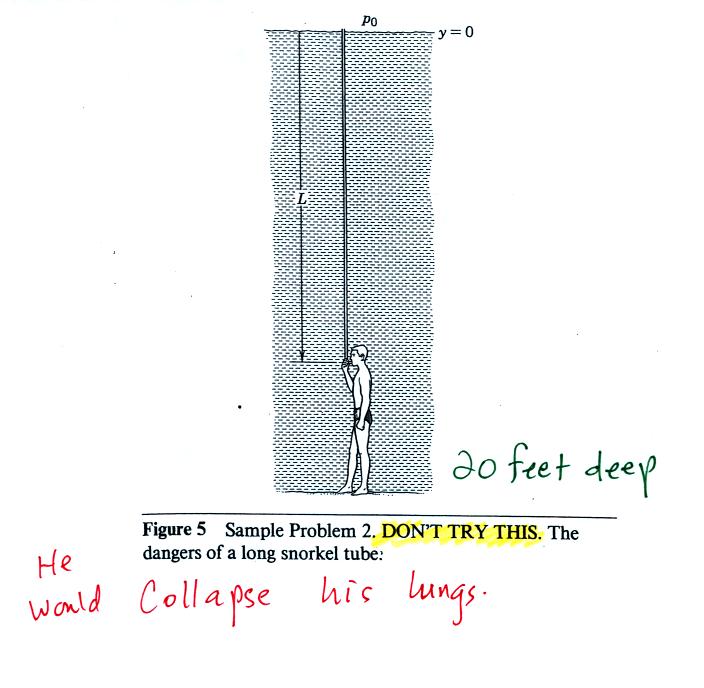
The person in the picture below (from a
Physics textbook) is 20 feet
underwater. At that depth there is a pretty large pressure
pushing against his body
from the surrounding water. The top of the snorkel is exposed to
the much lower air pressure at the top of the pool. If the
swimmer puts his mouth on the snorkel the pressure at the bottom of the
pull would collapse his lungs.
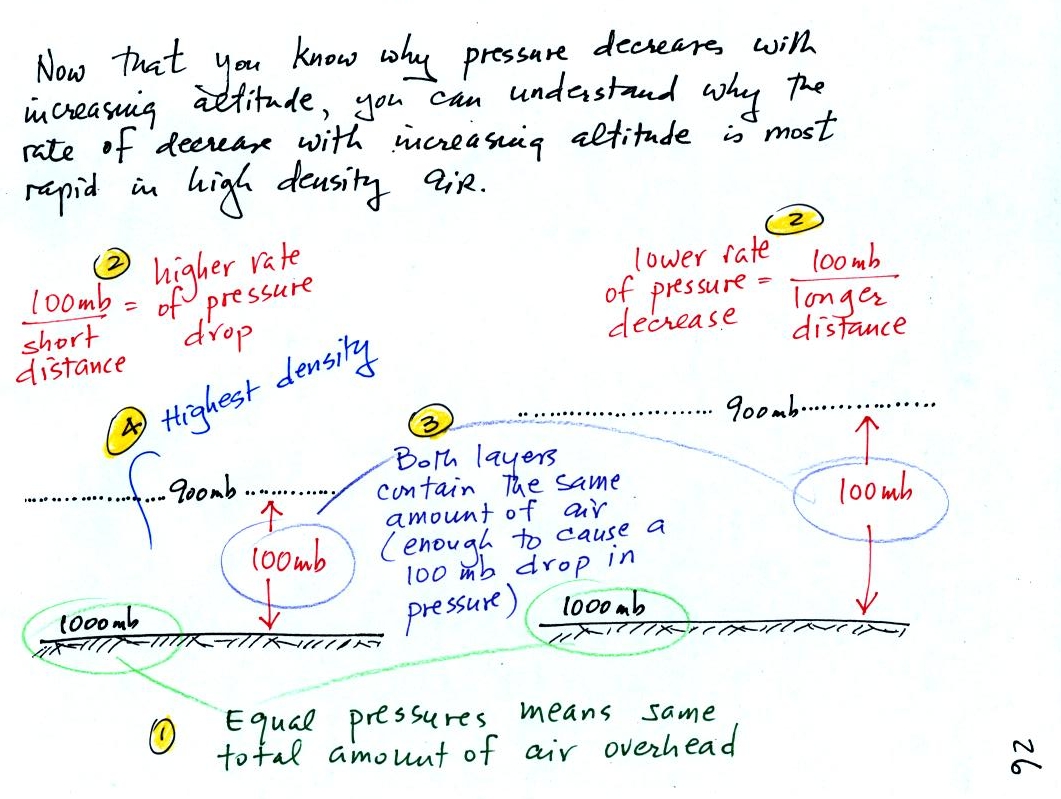
There is a lot going on in this
picture. 1000 mb at Point
1 is a typical value for sea level pressure. The fact that
the
pressures are equal at the bottoms of both
sides of the picture means that the weight of the atmosphere at the
bottom of the
picture on the left is the same as the weight of the atmosphere at the
bottom of the picture at right. The only way this can be true is
if there is the same total amount (mass) of air in both cases.
Point 2 - Moving upward from the ground we find that pressure
decreases
to 900 mb at the level of the dotted line in the picture at left.
This is what you expect, pressure decreases with increasing
altitude. In the figure at right you need to go a little bit
higher for the same 100 mb decrease.
The most rapid rate of pressure decrease with increasing
altitude is occurring in the picture at left because the 100 mb change
occurs in a shorter distance.
Point 3 -
Since there is a 100 mb drop in both the layer at left and
in
the
layer at right, both layers must contain the same amount (mass) of air.
Point 4 - The air in the picture at left is squeezed into a
thinner
layer than in the picture at right. The air density in the left
layer is higher than in the layer at right.
By carefully analyzing this figure we have proved to ourselves
that the
rate of pressure decrease
with altitude is higher in dense air than in lower
density air.
This is a fairly subtle but important concept. This concept
will come up 2 or 3 more times later in the semester. For
example, this concept partly explains why hurricanes can intensify and
get as
strong as they do.