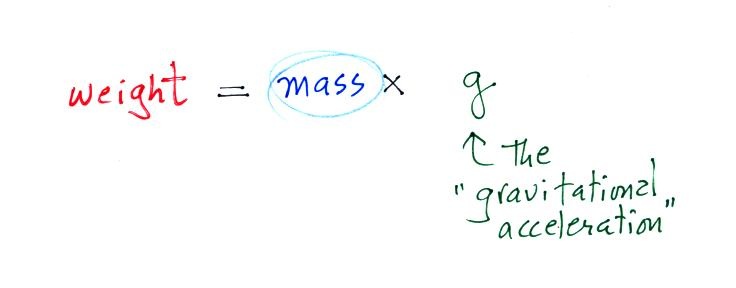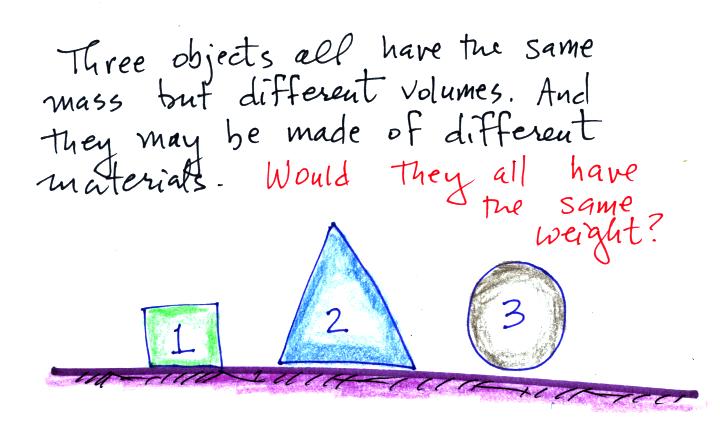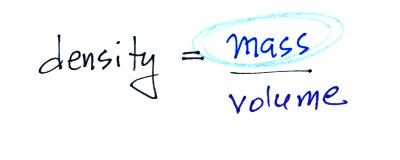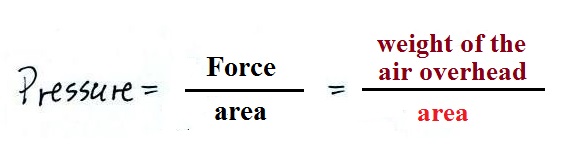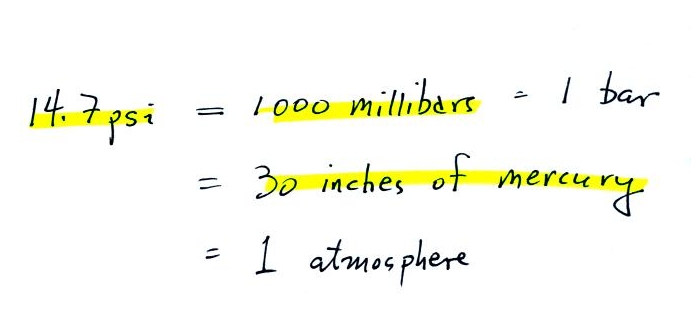Tuesday, Sep. 4, 2018
Another new discovery: Tash Sultana "Yin Yang"
(5:18), "Jungle"
(11:20)
We'll be using page 13c,
page 23a, page 23b, page 23c, page 23d, and page 24a from the
ClassNotes packet in today's class.
Particulate matter (PM)
We'll finish up our coverage of particulate matter today.
Sources of particulate
matter
Particulate matter can be produced naturally (wind blown dust,
clouds above volcanic eruptions, smoke from lightning-caused
forest and brush fires). Many human activities also produce
particulates (automobile exhaust for example). Gases
sometimes react in the atmosphere to make small drops or particles
(this is what happened in the photochemical smog
demonstration). Just the smallest, weakest gust of wind is
enough to keep these small particles suspended in the atmosphere.
A recent study estimates that more than 3.2 million people die
each year across the globe because of exposure to unhealthy levels
of PM25 (click here
to see a summary and some discussion of the study and here
to see the study itself). PM25 refers to particles with
diameters of 2.5 micrometers (µm) or less;
particles this small can penetrate deeply into the lungs.
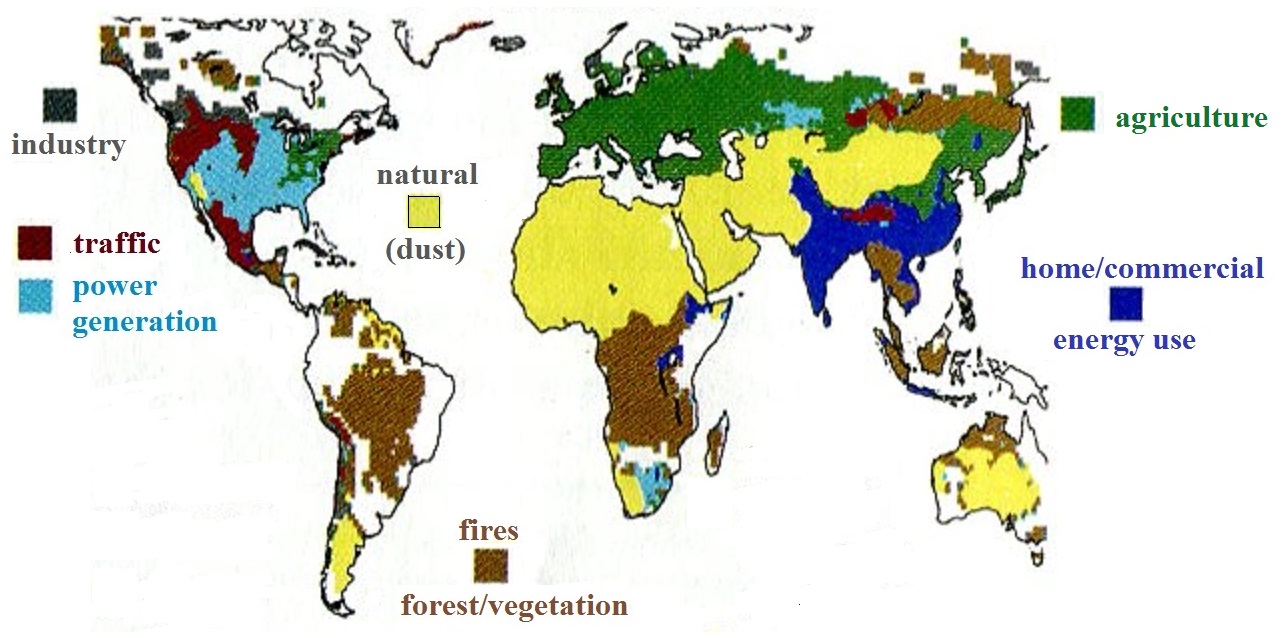
The study also attempted to determine the sources of the PM25
pollution. The figure below summarizes their
findings. Information like
this is important because you need to know what is adding
particulate matter to the air if you want to try and reduce
emissions.
CBS news has ranked the 30 cities in the world with
the most polluted air (based on World Health Organization
data for 2016) (https://www.cbsnews.com/pictures/the-most-polluted-cities-in-the-world-ranked/
). The report is interesting because there is a
photograph of each location and more detailed information
about the source of the pollution. Here is some of what
was mentioned: sandstorms, vehicle exhaust, aluminum
production, deforestation, burning waste, coal burning power
plants, oil production, leather tanning, brick factories,
chemical factories, burning tires to extract iron, steel
mills. Cities in China, India, Pakistan, Iran, and Saudi
Arabia appear multiple times in the list.
This map shows where some of the most polluted
places on earth are located (PM25 pollution) and comes from a
World Health Organization report "Exposure to ambient air
pollution from particulate matter for 2016" (http://www.who.int/airpollution/data/AAP_exposure_Apr2018_final.pdf?ua=1).
Effects of particulate matter on health
One of the main concerns with
particulate pollution is that the small particles might be a
health hazard ( a health advisory is sometimes issued during windy
and dusty conditions in Tucson).
Particles with dimensions of 10 µm
and less can be inhaled into the lungs (larger particles get
caught in the nasal passages). These inhaled particles
may be poisonous, might cause cancer, damage lung tissue, or
aggravate existing respiratory diseases. The smallest
particles can pass through the lungs and get into the blood
stream (just as oxygen does) and damage other organs in the
body.
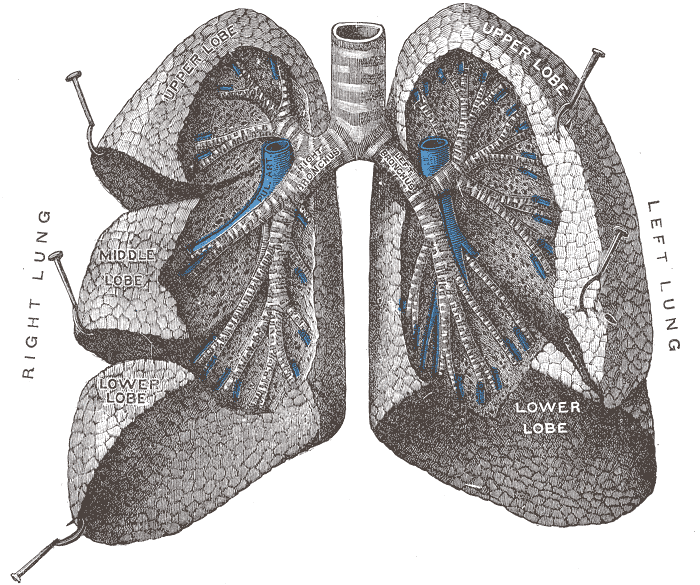
The figure below identifies some of the parts of the human
lung. The key point is that the
passageways get smaller and smaller as you move deeper into
the lungs. The smallest particles are the most dangerous
because they can penetrate furthest into the lungs.
The 2008 Summer
Olympics were held in Beijing and there was some concern
that the polluted air would affect the athletes
performance. Chinese authorities restricted
transportation and industrial activities before and during
the games in an attempt to reduce pollutant
concentrations. Rainy weather during the games may
have done the greatest amount of good.
Clouds and precipitation are the best way of cleaning
pollutants from the air. We'll learn later in the semester
that cloud droplets form on small particles in the air called
condensation nuclei. The cloud droplets then form raindrops
and fall to the ground carrying the particles with them.
The second main concern with particulates is the
effect they may have on visibility.
Here's a view of the Catalina mountains taken from the Gould
Simpson Building on the south side of campus.
Some rainy weather that had occurred just a day to two before the
photograph was taken had done a good job of cleaning the air and
the visibility was very good.
Windy weather a few days later
stirred up a lot of dust that was carried into town.
This picture was taken the day after the windy weather.
There is still a lot of fine dust particles in the air and the
visibility is pretty bad. Again the drop in
visibility is a consequence of light scattering. This is
part of what is examined in a new
1S1P topic.
We will look at some photographs from Beijing
(if that link doesn't work try this
one) where particulate pollution can be quite severe.
Here are some pictures from Harbin,
China (October, 2013). That's about as bad as visibility can
get, visibility in some cases is just a few 10s of feet. The
problem is limited to China, here's a picture from Paris
(March, 2014) and India
(November, 2017).
Just a week or two ago, smoke from forest fires in British
Columbia (Canada) was blown southward into Vancouver, Seattle, and
Portland (and smaller cities in Washington and Oregon). The
air quality was for a few days as bad as you'll find in the most
polluted cities in the world. (see "Wildfire Smoke Makes
Seattle and Portland World's Dirtiest Cities" published online by
National Geographic (https://www.nationalgeographic.com/environment/2018/08/news-seattle-portland-dirtier-air-quality-than-parts-of-asia/).
|

|
These two
photograph are from the National Geographic article
referenced above.
|
Smoke from fires in California will often be seen in Tucson.
Smoke from Canada and the Pacific Northwest does also sometimes
move into our area.
Satellite photograph taken early in the Fall 2017 semester
(with the new GOES16 satellite) showing smoke from wildfires
burning in Washington, Oregon, Idaho and Montana being carried
across much of the continental US (Hurricane Harvey is also
shown). Smoke from these fires made it into
southern Arizona where, at times, it had a noticeable effect on
visibility.

|

|
Photograph taken Saturday Aug. 26, 2017
when the air was free of smoke and visibility was pretty
good.
|
Photograph taken Tuesday on Aug. 29,
2017 when smoke from the fires in the Pacific northwest
was present. There has been a noticeable drop in
visibility.The camera was tilted down slightly in this
picture but the field of view is the same as the other
photograph.
|
Now we'll
change abruptly to an entirely different topic.
Mass,
weight, density, and pressure.
Pressure,
especially, is a pretty important
concept. Differences in atmospheric
pressure create winds and cause storms.
To better understand pressure we need to
review mass and weight.
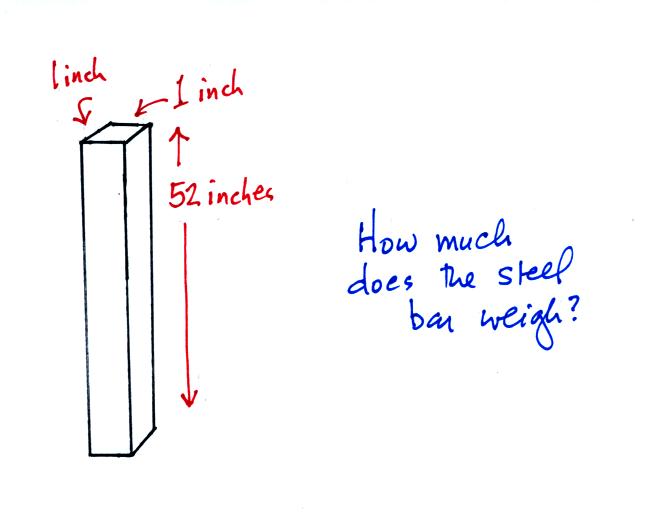
Weight is something you can feel. I'll
pass an iron bar around in class (it's
sketched below) - lift it and try to guess
it's weight. The fact that it is a 1" by
1" is significant. More about the bar
later in today's notes.
I used to
pass around a couple of small plastic bottles (see
below). One contained some water, the other an equal
volume of mercury (here's the source
of the nice photo of liquid mercury below at
right). I wanted you to appreciate how much
heavier and denser mercury is compared to
water.
But the plastic bottles have a way of getting brittle with
time and if the mercury were to spill in the classroom the
hazardous material people would need to come in and clean it
up. That would probably take a few days, would be very
expensive, and I would get into a lot of trouble. So
this semester I'll pass around a smaller, much safer, sample
of mercury so that you can at least see what mercury it looks
like (it's a recent purchase from a company in London).
I'll keep the plastic bottles of mercury up at the front of
the room just in case you want to see how heavy the stuff is.
It
isn't so much the liquid mercury that is a hazard, but
rather the mercury vapor. Mercury vapor is used in
fluorescent bulbs (including the new energy efficient CFL
bulbs) which is why they need to be disposed of
carefully. That is a topic that will come up again
later in the class.
Mercury and bromine are the only
two elements that are found naturally in liquid
form. All the other elements are either gases or
solids.
I am hoping that you will remember and understand the
following statement
atmospheric
pressure at any level in the atmosphere
depends on (is determined by)
the weight
of the air overhead
We'll
first review the concepts of mass, weight, and density
but understanding pressure is our main goal.
I've numbered the various sections to help with
organization. There's a summary at the end of
today's notes.
1.
weight
This is a good place to start because this is
something we are pretty familiar with. We
can feel weight and we routinely measure weight.
A person's weight also depends
on something else.
In outer space away from
the pull of the earth's gravity people are weightless.
Weight depends on the person and on the pull of gravity.
We
measure weight all the
time. What units
do we use?
Usually pounds, but
sometimes ounces or
maybe tons.
Students sometimes
mention Newtons, those
are metric units of
weight (force).
2. mass
Rather than just saying the
amount of something it is probably better to use the
word mass
It would be possible to have equal volumes of
different materials or the same total number of atoms or
molecules or two different materials, and still have different
masses.
Grams (g) and kilograms (kg) are commonly used units of
mass (1 kg is 1000 g).
3. gravitational
acceleration
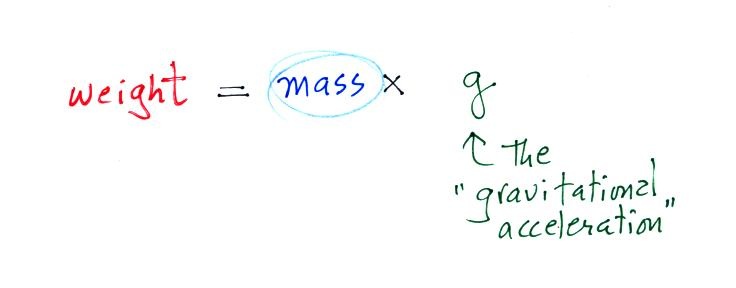
On the surface of the earth, weight is
mass times a constant, g, known as the
gravitational acceleration. The value of g
is what tells us about the strength of gravity on the earth;
it is determined by the size and mass of the earth. On
another planet the value of g would be
different. If you click here
you'll find a little (actually a lot) more information about
Newton's Law of Universal Gravitation. You'll see how
the value of g is determined and why it is called
the gravitational acceleration. These aren't details
you need to worry about but they're there just in case
you're curious.
Here's a question to test your understanding.
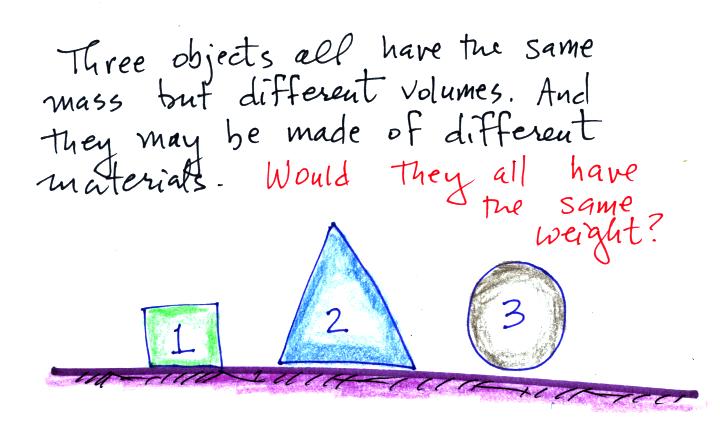
The masses are all the same. On the earth's surface the
masses would all be multiplied by the same value of g.
The weights would all be equal. If
all 3 objects had a mass of 1 kg, they'd all have a weight of 2.2
pounds. That's why we can use
kilograms and pounds interchangeably.
The following figure show a situation where two
objects with the same mass would have different weights.
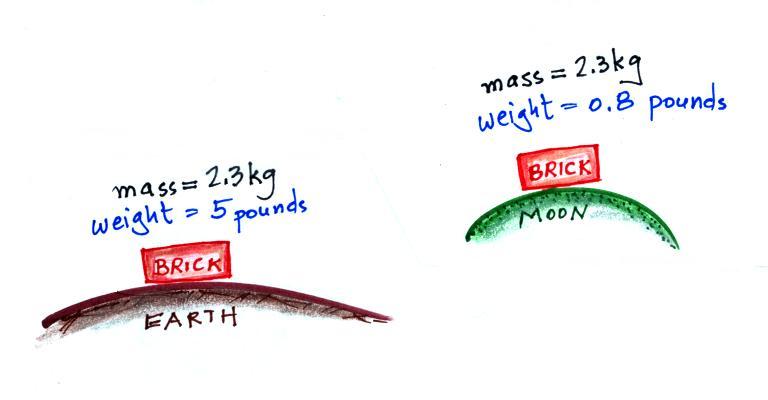
On the earth a brick has a mass of about
2.3 kg and weighs 5 pounds. If you were to travel to the
moon the mass of the brick wouldn't change (it's the same
brick, the same amount of stuff). Gravity on the moon is
weaker (about 6 times weaker) than on the earth because the
moon is smaller and it has less mass, the value of g
on the moon is different than on the earth. The brick
would only weigh 0.8 pounds on the moon.
The brick would weigh almost 12 pounds on the surface on
Jupiter where gravity is stronger than on the earth.
On the moon, a brick would have the same mass, the same
volume, the same density, but a different weight as(than)
it would on the earth.
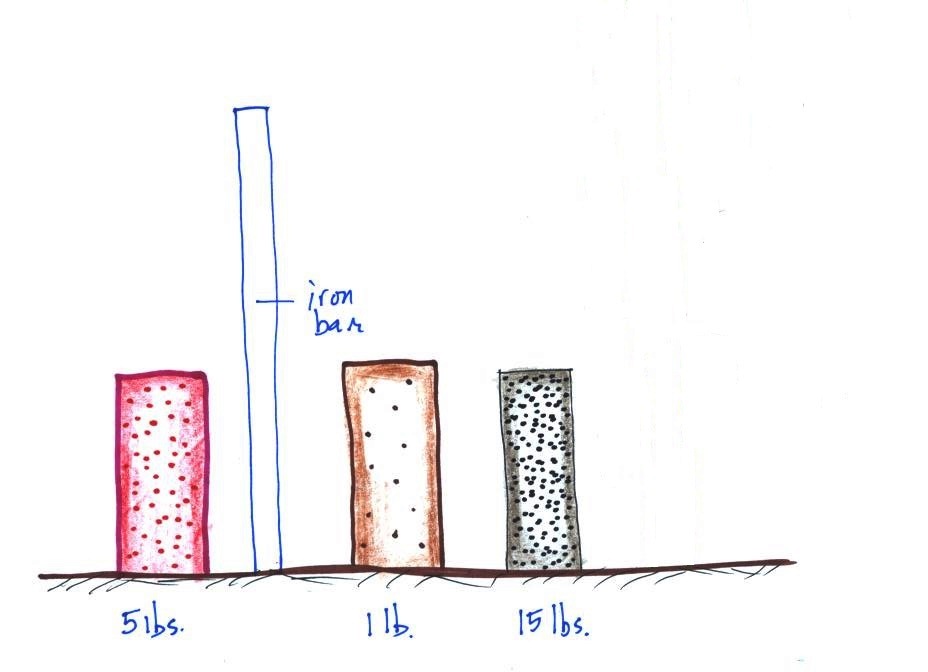
The three objects below
were not passed around class (one of them is pretty
heavy). The three objects all have about
the same volumes. One is a piece of wood,
another a brick, and the third is something
else.
The
easiest way to determine which is which is to lift each
one. One of them weighed about 1 pound (wood), the 2nd
about 5 pounds (a brick) and the last one was 15 pounds (a
block of lead).
The point of all this was to get you thinking about
density. Here we had three objects of about
the same size with very different weights. Different
weights means the objects have different masses (since weight
depends on mass). The three different masses, were
squeezed into roughly the same volume producing objects of
very different densities.
4. density
The brick is in the back, the lead
on the left, and the piece of wood (redwood) on the right.
The wood is less dense than water (see the table below) and
will float when thrown in water. The brick and the lead
are denser than water and would sink in water.
We'll be more concerned with air in this
class than wood, brick, or lead.
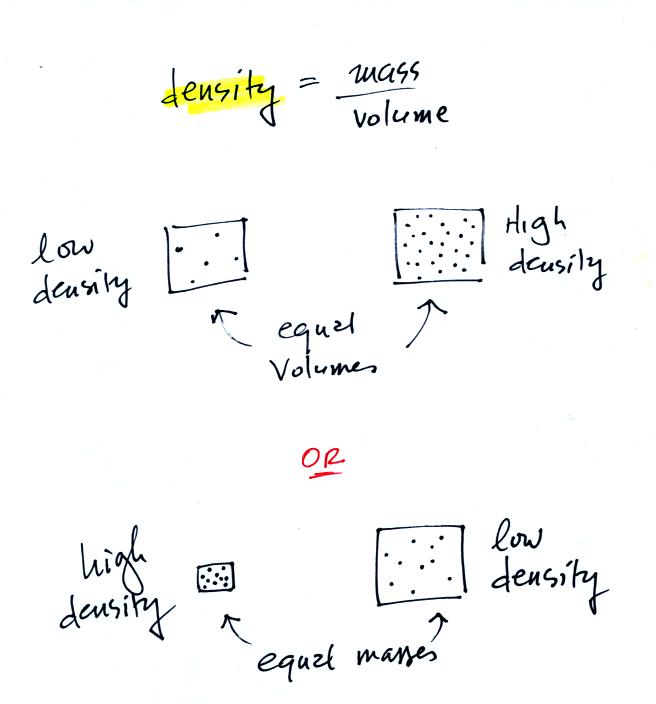
In the first example
below we have two equal volumes of air but the amount in
each is different (the dots represent air
molecules).
The amounts of air (the masses) in the second example are the
same but the volumes are different. The left example
with air squeezed into a smaller volume has the higher
density.
material
|
density g/cc
|
air
|
0.001
|
redwood
|
0.45
|
water
|
1.0
|
iron
|
7.9
|
lead
|
11.3
|
mercury
|
13.6
|
gold
|
19.3
|
platinum
|
21.4
|
iridium
|
22.4
|
osmium
|
22.6
|
g/cc = grams per cubic centimeter
cubic centimeters are units of volume - one cubic
centimeter is about the size of a sugar cube
1 cubic centimeter is also 1 milliliter (mL)
I would sure like to get my hands on a brick-size
piece of iridium or osmium just to be able to feel how
heavy it would be - it's about 2 times denser than
lead.
Here's a more subtle concept. What if you were in outer
space with the three wrapped blocks of lead, wood, and
brick? They'd be weightless.
Could you tell them apart then? They would still have very
different densities and masses but we wouldn't be able to feel how
heavy they were.
5.
inertia
I think the following illustration will
help you to understand inertia.
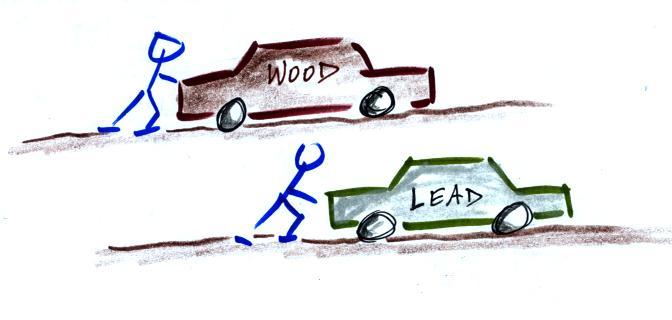
Two stopped cars. They are the same size except
one is made of wood and the other of lead. Which
would be hardest to get moving (a stopped car resists
being put into motion). It would take considerable
force to get the lead car going. Once the cars are
moving they resist a change in that motion. The
lead car would be much harder to slow down and stop.
This is the way you could try to distinguish
between blocks of lead, wood, and brick in outer space.
Give them each a push. The wood would begin moving more
rapidly than the block of lead even if both are given
the same strength push.
I usually
don't mention in class that this concept of
inertia comes from Newton's 2nd law of motion
F = m a
force = mass x acceleration
We can rewrite the equation
a = F/m
This shows cause and effect more clearly. If you exert a
force (cause) on an object it will accelerate (effect).
Acceleration can be a change in speed or a change in direction (or
both). Because the mass is in the denominator, the
acceleration will be less when mass (inertia) is large.
Here's where we're at
From left to right the brick, the iron bar, the piece
of wood, and the lead block. They're all standing on end.
The weight of the iron bar is still unknown.
Now
we're close to
being ready to
define (and
hopefully
understand)
pressure.
It's a pretty
important
concept.
A lot of what
happens in the
atmosphere is
caused by
pressure
differences.
Pressure
differences
cause
wind.
Large pressure
differences
(such as you
might find in
a tornado or a
hurricane) can
create strong
and
destructive
storms.
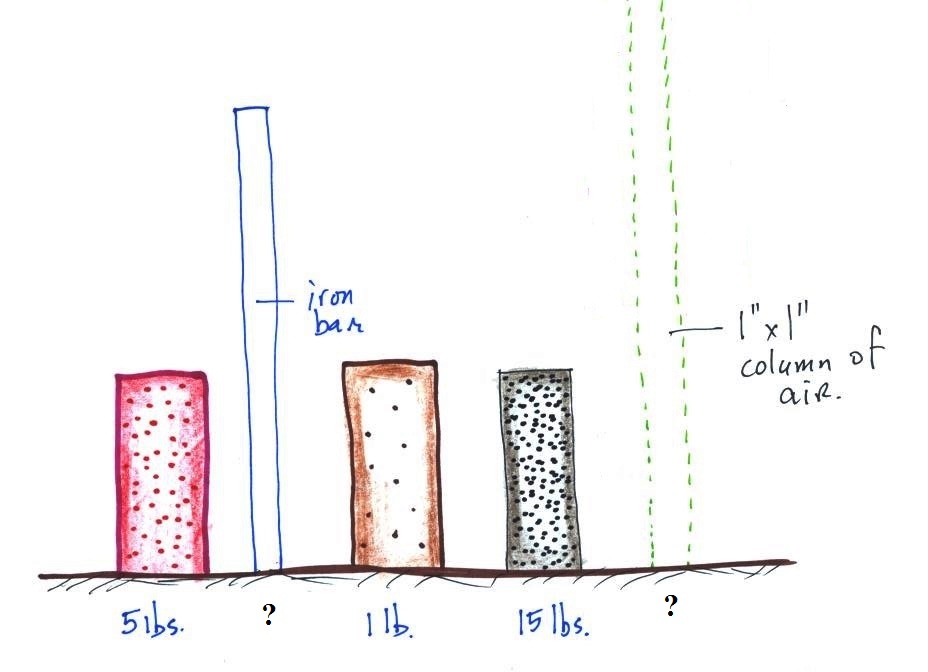
The air that surrounds the earth has mass. Gravity pulls
downward on the atmosphere giving it weight. Galileo
conducted a
simple experiment to prove that air has weight (in the
1600s).
We
could add a very
tall 1 inch x 1
inch column of air
to the
picture.
Other than being a
gas, being
invisible, and
having much lower
density it's
really no
different from the
other objects.
6. pressure
Atmospheric pressure at
any level in the atmosphere
depends on (is determined
by)
the weight of the air
overhead
This
is one way, a sort of large, atmosphere size-scale
way, of understanding air pressure.
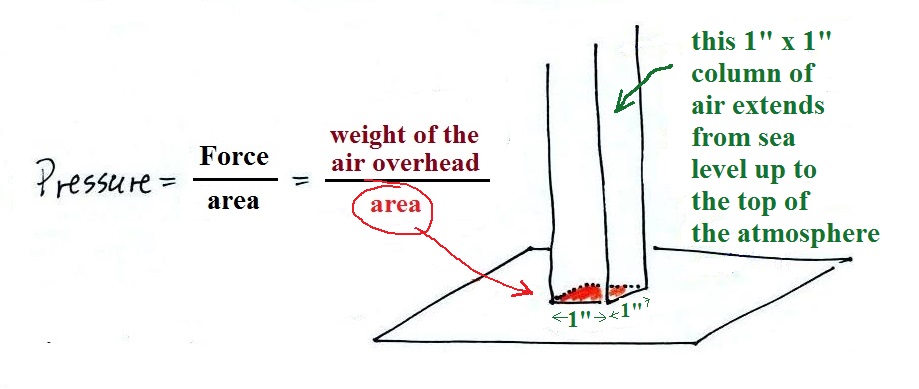
Pressure depends on, is determined by, the weight of the
air overhead. To determine the pressure you need to
divide by the area the weight is resting on.
and here we'll apply the
definition to a column of air stretching from sea
level to the top of the atmosphere (the figure below
is on p. 24 in the ClassNotes)
Pressure is defined as force divided by area. Atmospheric
pressure is the weight of the air column divided by the area at
the bottom of the column (as illustrated above).
Under normal conditions a 1 inch by 1 inch column of air
stretching from sea level to the top of the atmosphere will weigh
14.7 pounds.
Normal atmospheric pressure at sea level is 14.7 pounds per square
inch (psi, the units you use when you fill up your car
or bike tires with air).
Now back to the iron bar. The bar actually weighs
14.7 pounds (many people I suspect think it's heavier than
that). When you stand the bar on end, the pressure at
the bottom would be 14.7 psi.
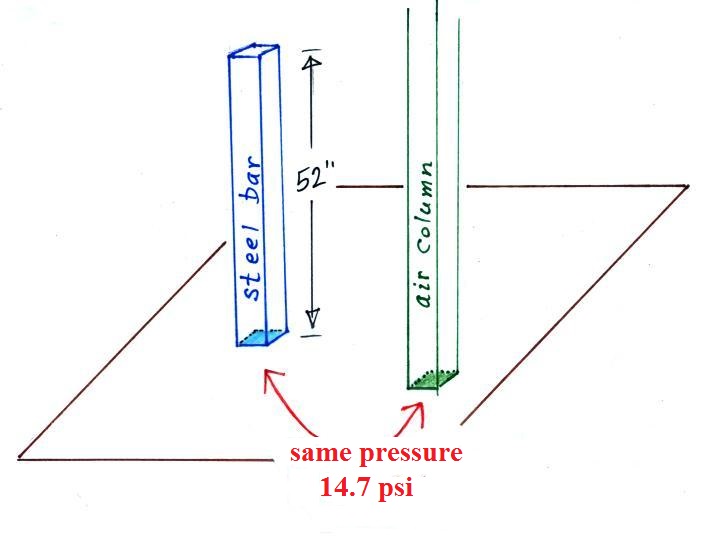
The weight of the 52 inch
long 1" x 1" steel bar is the same as a 1" x 1" column
of air that extends from sea level to the top of the
atmosphere 100 or 200 miles (or more) high. The
pressure at the bottom of both would be 14.7 psi.
7. pressure units
Pounds per square inch, psi, are
perfectly good pressure units, but they aren't the ones
that meteorologists use most of the time.
Typical sea
level pressure is 14.7 psi or about 1000 millibars
(the units used by meteorologists and the units that we will
probably mostly use in this class) or about 30 inches of
mercury. Milli means 1/1000 th. So
1000 millibars is the same as 1 bar. You sometimes see
typical sea level pressure written as 1 atmosphere.
Here's a summary of the material covered in the second portion
of today's class.