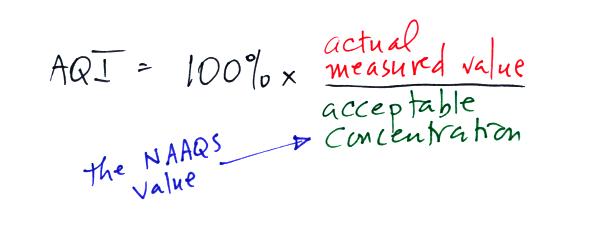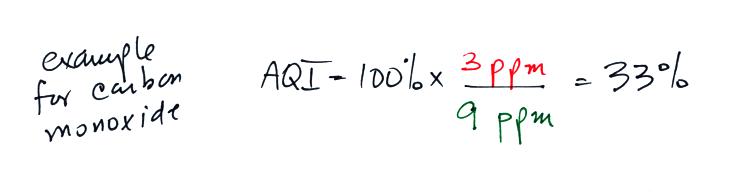Monday August 29, 2011
click here to
download today's notes in a more printer friendly format
Three songs from Julia Lee
before class this afternoon ("Gotta Gimme What'cha
Got", "Don't Come
Too Soon" and "Ain't It A Crime" )
The 1st Optional
Assignment of the semester was handed out in
class today. It's due at the start of class on Friday. You
can earn extra credit points on the optional assignment. But you
should finish the assignment before coming to class, don't let me find
you
furiously trying to finish the assignment before class on Friday.
You can always turn in assignments ahead of time.
A little more information on carbon monoxide that we need to finish
before a short demonstration and starting a section on sulfur dioxide.
Here's a summary of what we've learned about carbon monoxide last
Friday.
Let's imagine that the average carbon monoxide concentration in Tucson
air yesterday was 3 ppm. Is this a high and unhealthy value or
was the air quality OK? We need some more information. We
need to know what an acceptable concentration level for carbon monoxide
is. The EPA has done just that ( a neater version of the top of
p. 8 in the photocopied ClassNotes is shown below)
The six main pollutants are listed above (there are many more).
The concentration of lead in air has decreased
significantly since lead was removed from gasoline (the
following quote is from a
Wikipedia
article
on
gasoline: "In the US,standards to phase out
leaded gasoline were first implemented in 1973 ..... In 1995, leaded
fuel accounted for only 0.6% of total gasoline sales ...... From 1
January 1996, the Clean Air Act banned the sale of leaded fuel
for use in on-road vehicles. Possession and use of leaded gasoline in a
regular on-road vehicle now carries a maximum $10,000 fine in the US.")
There was a question at about this point about
chlorofluorocarbons. CFCs are largely not toxic. The
molecules are very stable and unreactive compounds, however, that can
make their way up into the stratosphere where they deplete
stratospheric ozone. For this reason, a ban on the use of CFCs as
a propellant in aerosol cans was put into place in the US in
1978. International agreements since then such as the 1987
Montreal Protocol have sought to drastically reduce and eventually
eliminate CFC production.
In Tucson,
carbon monoxide, ozone, and particulate matter are of primary concern
and daily measurements are reported in the city newspaper. Rather
than report the actual measured values, an Air Quality Index value is
reported instead. The AQI is the ratio of the
measured to accepted
concentrations multiplied by 100%.
If we plug in the 3 ppm value mentioned above for carbon monoxide,
the AQI value would be
The air quality in this case would be good. Air becomes
unhealthy when the
AQI value exceeds 100%. The units "ppm", by the way,
stands for "parts per million." A CO concentration of 3 ppm would
mean that in 1 million air molecules 3 of them would be carbon monoxide.
This information is found on the bottom of p. 8 in the photocopied
ClassNotes. Current
Air
Quality
Index
values
for
Tucson
are
available online.
Carbon monoxide is a serious
hazard indoors where is can build to much higher levels than would ever
be found outdoors. This next link is to a newspaper article
describing an
incident at Virginia Tech (that occurred near the beginning of the
school year in 2007). Carbon
monoxide
from a malfunctioning hot water heater sickened 23 Virginia Tech
students in an apartment complex. The CO concentration is
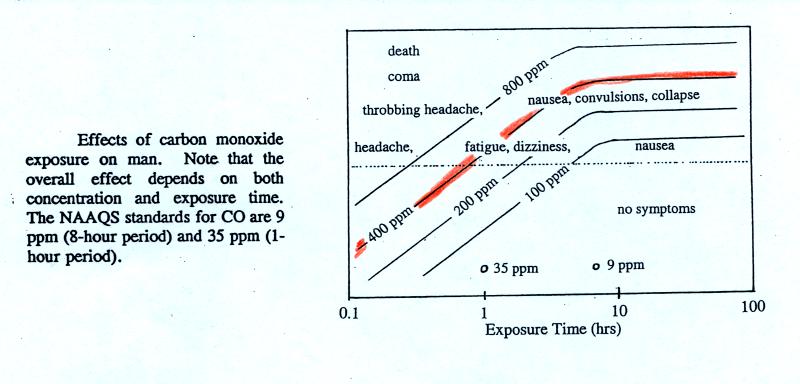
thought to have reached 500 ppm. You can get an idea of what
kinds of health effects concentrations this high could cause from the
figure. on p. 9 in the photocopied ClassNotes.
The 400
ppm line in the ClassNotes approaches the level where CO would cause
coma and
death. At Virginia Tech several students were found unconscious
and one or two had stopped breathing but they were revived.
Carbon monoxide
alarms are relatively inexpensive (~$50) and readily available at most
hardware
stores. They will monitor CO concentrations indoors and warn you
when
concentrations reach hazardous levels. Indoors CO is
produced by gas furnaces and water heaters that are
either operating improperly or aren't being adequately vented
to the outdoors. A few hundred people are killed indoors by
carbon
monoxide every
year in the United States. You can learn
more about carbon monoxide hazards and risk prevention at the Consumer Product
Safety Commission web page.
You might have missed this
link in last Friday's lecture notes.
This was a good point for a demonstration, one that was once voted
the prettiest demonstration of the semester.
You are able to see a lot of things in the atmosphere (clouds,
fog, haze, even the blue sky) because of scattering of light. I'm
going to try to make a cloud of smog in class later this week.
The
individual droplets making up the smog cloud are too small to be seen
by
the
naked eye. But you will be able to see that they're there because
the droplets scatter light. So we took some time for a
demonstration that tried to show you
exactly what light scattering is.
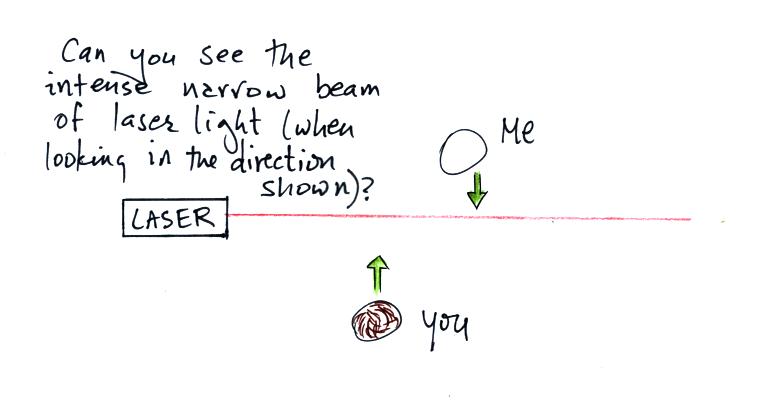
In the first part of the demonstration a narrow beam of intense
red
laser light was shined from one side of the classroom to the
other.
Looking down on the situation in the figure above. Neither
the students or the instructor could see the beam of light.
Nobody could see the beam because there weren't any rays of light
pointing from the laser beam toward the students or toward the
instructor.
The instructor would have been
able
to see the beam if he had stood at the end of the beam of laser light
and looked back along the beam of light toward the laser. That
wouldn't have been a smart thing to do, though, because the beam was
strong
enough to possibly damage his eyes (there's a warning on the
side of the laser).
Everybody was able to see a bright red spot where the laser beam struck
the wall.
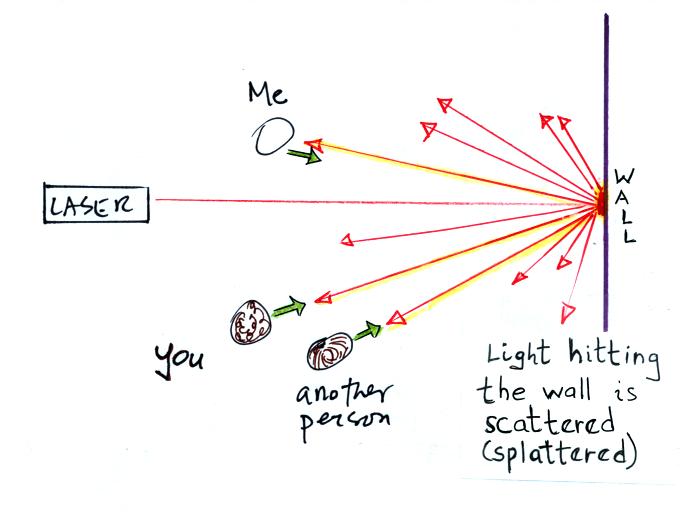
This is because when the intense beam of
laser light
hits the wall it
is scattered (splattered is a
more descriptive term). The original beam is broken up into a
myriad of weaker rays
of light that are sent out in all directions. There is a ray of
light
sent in the direction of every student in the class. They see the
light because they are looking back in the direction the ray came
from. It is safe to look at this light because the original
intense beam is split up into many much weaker beams.
Next we clapped some erasers together so that some small
particles of chalk dust fell into the laser beam.
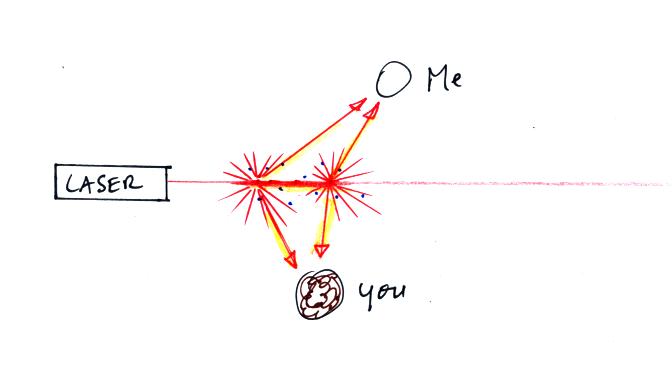
Now instead
of a single spot on the wall, students
saws lots of
points of light coming from different positions along a straight
segment of the laser
beam. Each of these points of light was a particle of chalk, and
each piece of chalk dust was intercepting laser light and sending light
out in all directions. Each student saw a ray of light coming
from
each of the chalk particles.
We use chalk because it is white, it will scatter rather
than absorb visible light. What would you have seen if black
particles
of soot had been dropped into the laser beam?
In the last part of the demonstration we made a cloud by
pouring some
liquid nitrogen into a cup of water. The cloud droplets are much
smaller than the chalk particles but are much more numerous. They
make very good scatterers.
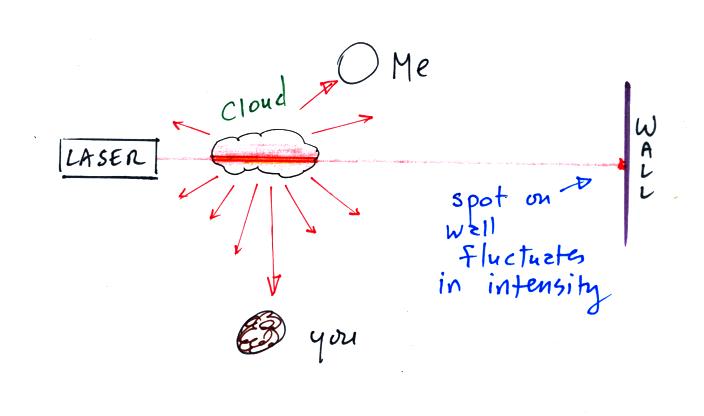
The laser beam is visible in the left 2/3 rds of the picture
because it is passing through cloud and light is being scattered toward
the camera. There wasn't any cloud on the right 1/3rd of the
picture so you can't see the laser beam over near Point 1.
There's something else going on in this picture also. We're
not just seeing the narrow beam of laser light but some of the cloud
outside the laser beam is also visible.
Up to this point we've just considered single scattering. A beam
of light encounters a cloud droplet or a particle of chalk and gets
redirected and then travels all the way to your eye or to a
camera. That's what's happening at Point 2. You just see
the narrow laser beam. But sometimes the scattered ray of light
runs into
something else and gets scattered again. This is called multiple
scattering. And that is what is illuminating the cloud alongside
the beam of laser light at Point 3. Light is first scattered by a
cloud droplet in the beam. As it leaves the beam it runs into
another droplet and gets scattered again. So now it looks like it
is coming from the cloud surrounding the laser beam rather than from
the beam itself.
Here's a comment that wasn't mentioned in
class
Air molecules are able
to scatter light too, just like cloud droplets. Air molecules are
much smaller than cloud droplets and don't scatter much light.
That's why you couldn't see the laser beam as it was traveling from one
side of the classroom to the other through the air. Outdoors we
are able to see sunlight scattered by air molecules. This is true
for a couple of reasons. The sunlight is much stronger than the
laser beam and its shining through a lot more air.
Sunlight is
white light which means it's made up of a mixture of violet, blue,
green, yellow, orange, and red
light. Air molecules have an unusual property: they scatter the
shorter wavelengths (violet, blue, green) much more readily than the
longer wavelength colors in sunlight (yellow, orange, and red).
When you look away from the sun and look at the sky, the blue color
that you see are the shorter wavelengths in sunlight that are being
scattered by air molecules.
We'll come back to the topic of light scattering later this week or
early next week. when we
cover particulate matter and its effect on visibility.
We had a little time to start a short section on sulfur dioxide, the
2nd air pollutant we will be concerned with.
The following information is on p. 11 in the photocopied ClassNotes.

Sulfur dioxide is produced by the
combustion of sulfur
containing
fuels such as coal. Combustion of fuel also produces carbon
dioxide and carbon monoxide. People probably first became aware
of sulfur dioxide because it has an unpleasant smell.
Carbon
dioxide and carbon monoxide are odorless. That is why sulfur
dioxide was the first pollutant people became aware of.
Volcanoes are a natural source of sulfur dioxide.
Sulfur dioxide has been involved in
some of the world's worst air pollution disasters. If not the
deadliest, The
Great
London Smog of 1952 is in the top three. Because the atmosphere
was stable, SO2
emitted into air
at ground level couldn't mix with cleaner air above. The SO2
concentration was able to build to dangerous levels. 4000 people
died during this 4 or 5 day period. As many as 8000 additional
people died in the following weeks and months.
Some
of the photographs below come from articles published in 2002 on the
50th anniversary of the event.
The sulfur dioxide didn't
kill people directly. Rather it would aggravate
an existing
condition of some kind. The SO2 probably
also made people susceptible
to bacterial
infections such as pneumonia. Here's
a
link that discusses the event and its health effects in more
detail.
London type smog which contains sulfur dioxide and is
most
common
during the winter is very different from photochemical or Los Angeles
type smog, something we will be learning about later this week.
Los
Angeles type smog contains ozone and is most
common in the summer.
Some other air pollution disasters also involved high SO2
concentrations. One of the deadliest events in the US occurred in
1948 in Donora, Pennsylvania.
"This eerie photograph was taken at noon on Oct.
29, 1948 in Donora, PA as deadly smog enveloped the town. 20 people
were asphyxiated and more than 7,000 became seriously ill during this
horrible event." The photograph below shows some of the mills
that were operating in Donora at the time. The factories were not
only emitted pollutants into the air but probably also discharging
pollutants into the river.
from: http://oceanservice.noaa.gov/education/kits/pollution/02history.html
from: http://www.eoearth.org/article/Donora,_Pennsylvania
"When Smoke Ran Like Water," a
book
about air pollution is among the books that you can check out, read,
and report on to fulfill part of the writing requirements in this class
(instead of doing an experiment report). The
author, Devra Davis, lived in Donora Pennsylvania at the time of the
1948 air pollution episode.