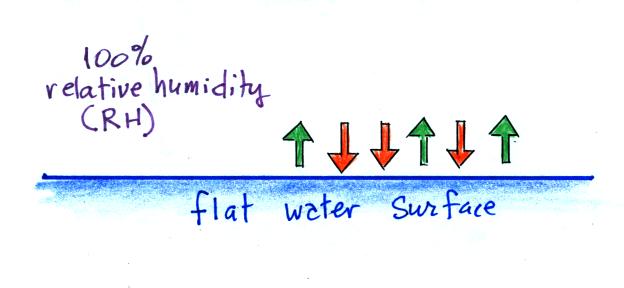
| it is much easier for water vapor to condense onto small particles called condensation nuclei |
rather than just
condensing and forming small droplets of pure water |

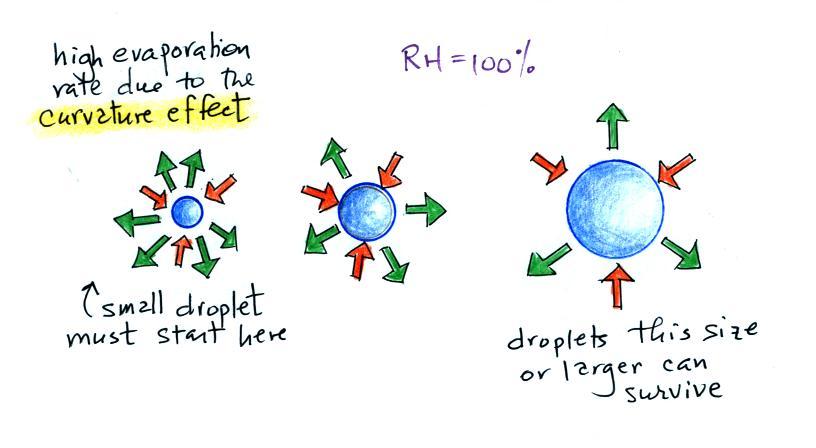
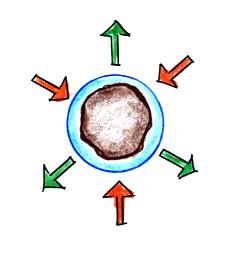

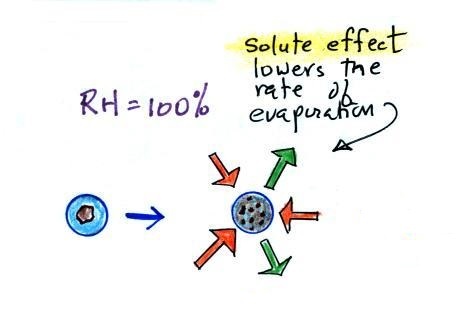 |
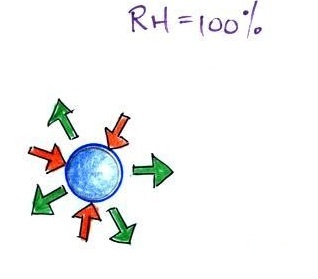 |
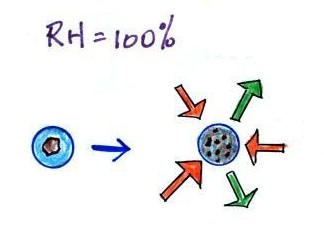
| solution droplet |
pure water droplet |
 |
 |
| the droplet is able to
grow |
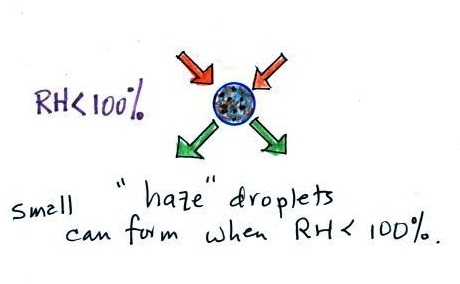
the droplet is in
equilibrium with its surroundings even when the RH is less than 100% |