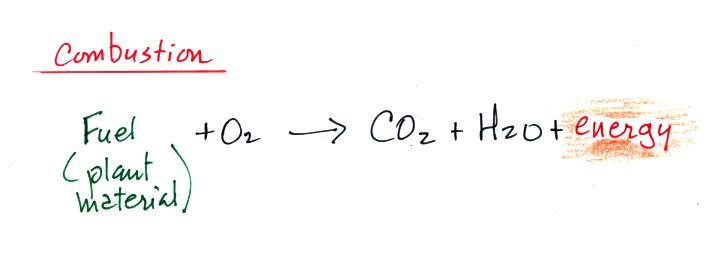The earth's original atmosphere and the origin(s) of
our present atmosphere
Our present day atmosphere (shown below at left) is very
different from the earth's original atmosphere (at right below)
which was mostly hydrogen and helium with lesser amounts of
ammonia and methane.
The early atmosphere either escaped into space (the earth
was hot and light weight gases like hydrogen and helium were
moving around with enough speed that they could overcome the
pull of the earth's gravity) or was swept into space by the solar wind
(click on the link if you are interested in learning more about
the solar wind, otherwise don't worry about it).
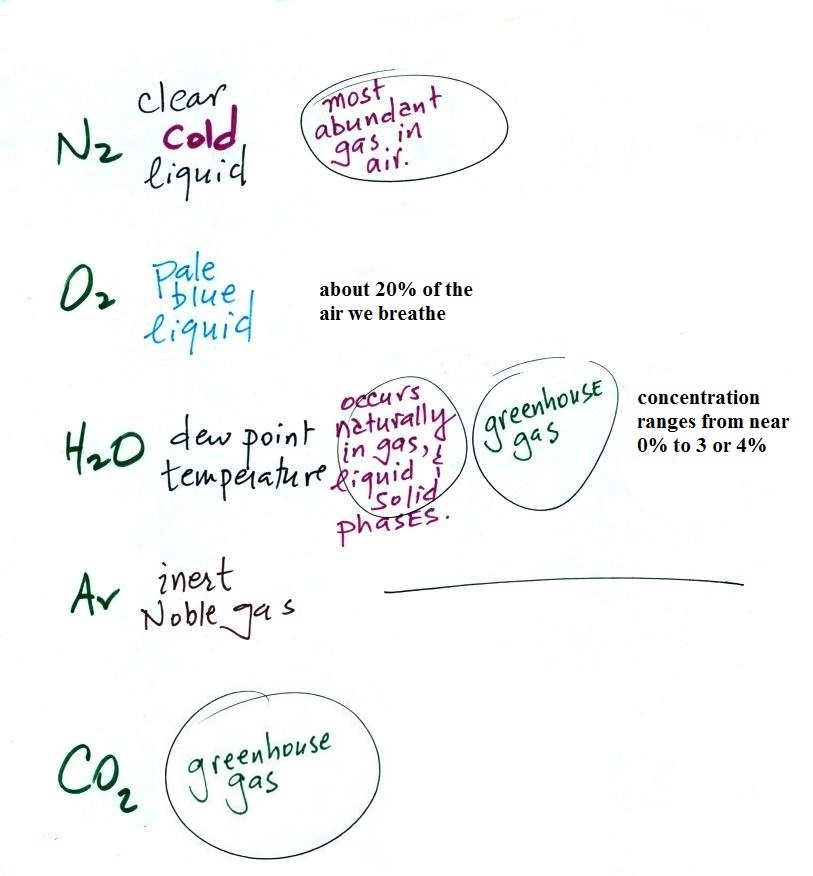
With the important exception of oxygen (and argon perhaps),
most of our present atmosphere is though to have come from
volcanic eruptions. In addition to ash, volcanoes send a lot
of water vapor, carbon dioxide, and sulfur dioxide into the
atmosphere. Carbon dioxide and water vapor are
two of the 5 main gases in our present atmosphere.
Volcanoes also emit lots of other gases,
many of them are very poisonous. Some of them are shown
on the right side of the figure. The gases in the "also"
list were mentioned in a lot of online sources, the gases in
the "perhaps" list were mentioned less frequently. The
relative amounts of these "also" and "perhaps" gases seems to
depend a lot on volcano type.
As the earth began to cool the water vapor condensed and
began to create and fill the earth's oceans. Carbon
dioxide dissolved in the oceans and was slowly turned into
rock. Nitrogen containing compounds like ammonia (
NH3 ) and
molecular nitrogen ( N2 ) are also emitted by
volcanoes. I'm guessing that the nitrogen in NH3
reacted with other gases
to produce N2.
Molecular nitrogen is pretty nonreactive so
once in the air its concentration was able to built up over
time.

The photo above shows the
Eyjafjallajokull volcano in Iceland photographed on Apr. 17,
2010 (image
source)
Here are some additional
pictures of the Eyjafjallajökull
volcano which you should really have a look
at. Eyjafjallajökull
caused severe disruption of airline travel between the US
and Europe. Here's
another
set of photos also from the Boston Globe.
This photograph is a mosaic of 4 images taken over a
roughly 20 minute period from an altitude of 28.6 km
(17.8 miles). Photo credit: "Comet 67P on 19
September 2014 NavCam mosaic" by ESA/Rosetta/NAVCAM
found a
Wikipedia article on the Rosetta mission.
|
A mosaic of the first two images showing the Philae
lander on the comet's surface. photo credit:
ESA/Rosetta/Philae/CIVA photo source
|
Even more amazing than the photographs of the Icelandic
volcano are these photographs of Comet
67P/Churyumov-Gerasimenko taken by the European Space Agency
Rosetta spacecraft. The spacecraft was launched on March
2, 2004 and went into orbit around the comet on August 6,
2014. On November 12, the Rosetta spacecraft deployed
the Philae lander which successfully landed on the surface of
the comet and operated for a brief time. The lander was
not able to fully deploy its solar panels and used up its
battery power and went into "sleep" mode after about 60 hours
of operation. In June this year the comet had
moved into a sunnier part of its orbit and the lander
began sending data again. The comet has just
reached perihelion (shortest distance between the comet and
the sun). The Rosetta spacecraft has photographed
material being ejected from the comet (you'll find a nice
animation here).
I've included this photograph of a comet because some
researchers don't believe that volcanic activity
alone would have been able to account for all the water that
is on the earth (oceans cover about 2/3rds of the earth's
surface). They believe that
comets and asteroids colliding with the earth may have brought
significant amounts of water. The Rosetta spacecraft has
determined that the water on this particular comet differs
from the composition of the water in the earth's oceans (this
reference reports "The ratio of deuterium to hydrogen in the water from the comet
was determined to be three times that found for terrestrial
water.") This suggests that comets like 67P were
probably not an important source of the earth's water.
Where did the oxygen in
our atmosphere come from?
Oxygen is in H2O, CO2,
and SO2 (and many of the other gases emitted by
volcanoes) but volcanoes aren't a direct source of the molecular
oxygen ( O2 )
that is present in air. Where did the
O2 come from? There
are a couple of answers to that question.
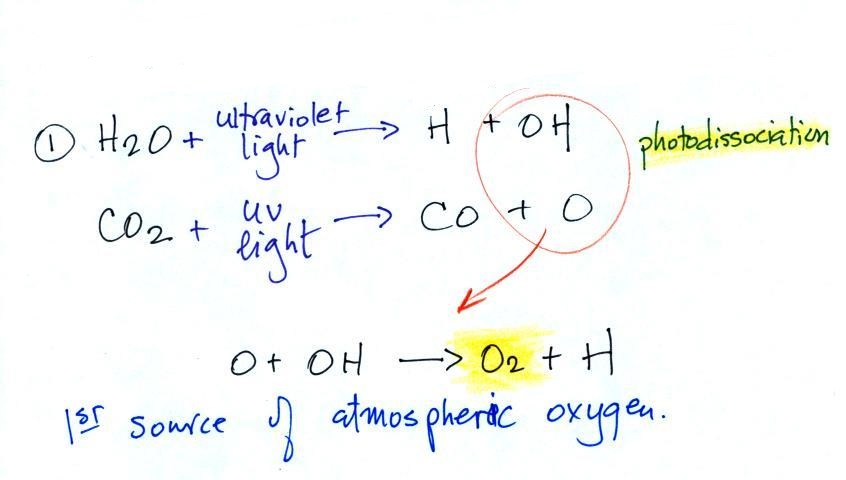
1st source of atmospheric oxygen
Oxygen is thought to have come from
photo-dissociation of water vapor and carbon dioxide by
ultraviolet (UV) light (the high energy UV light is able to split
the H20 and CO2 molecules into pieces). Two of the pieces,
O and OH, then react to form O2 and H.
By the way I don't expect you to remember the chemical formulas
in the example above. It's often easier and clearer to show
what is happening in a chemical formula than to write it out in
words. If I were to write the equations down, however, you
should be able to interpret them. Ultraviolet is a
dangerous, high energy, potentially deadly form of light and it's
probably also good to remember that ultraviolet light is capable
of breaking molecules apart.
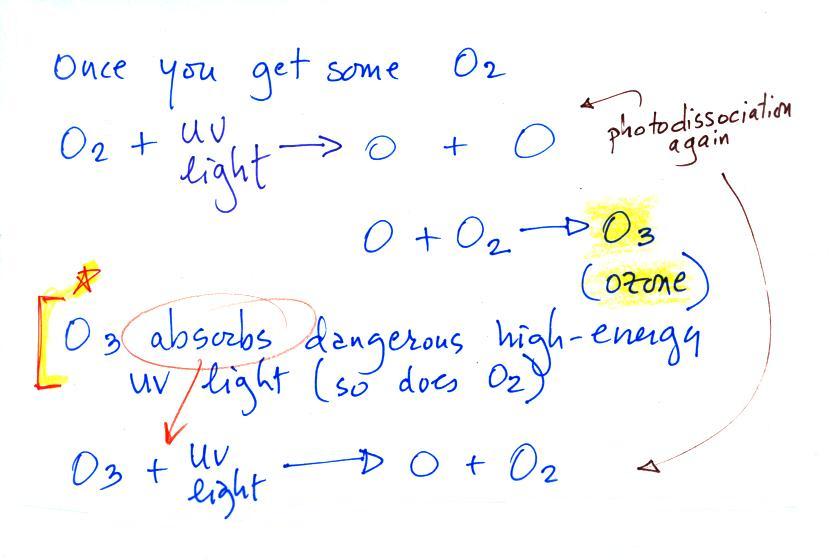
Once molecular oxygen (O2) begins to accumulate in
the air UV light can split the O2 apart to make atomic oxygen
(O). The atoms of oxygen can react with molecular oxygen
to form ozone (O3).
Ozone in the atmosphere began to absorb the dangerous and
deadly forms ultraviolet light and life forms could then begin
to safely move from the oceans onto land (prior to the buildup
of ozone, the ocean water offered protection from UV
light. A molecule of O3 absorbs some UV
preventing it from reaching the ground.
O3 + UV light ---> O2 + O
You might think the O2
and O would recombine. But if you picture hitting
something with a hammer and breaking it, the pieces usually fly
off in different directions. That's essentially what
happens with the O and O2 .
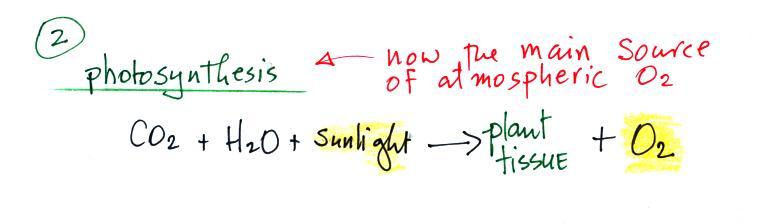
2nd and more important source of atmospheric
oxygen.
Once plant life had developed sufficiently and once plants moved
from the oceans onto land, photosynthesis became the
main source of atmospheric oxygen.
Photosynthesis in its most basic form is shown in the chemical
equation above. Plants need water, carbon dioxide, and
sunlight in order to grow. They can turn can turn H20 and CO2 into plant material. Photosynthesis
releases oxygen as a by product.
Combustion is really just the opposite of photosynthesis and is
shown below.
We burn fossil fuels (dead but undecayed plant material) to
generate energy. Water vapor and carbon dioxide are by
products. Combustion is a source of CO2 (photosynthesis is
a "sink" for atmospheric CO2 , it removes CO2 from the air).
Carbon dioxide is the subject of an upcoming 1S1P assignment
and we'll see these two equations again there and when we study
the greenhouse effect and global warming.
Here's a detail that I often forget to
mention when this material is covered in class (and
something you probably don't need to remember). The argon we
have in the atmosphere apparently comes from the radioactive decay
of potassium in the ground. Three isotopes of
potassium occur naturally: potassium-39 and potassium-41 are
stable, potassium-40 is radioactive and is the source of the argon
in the atmosphere.
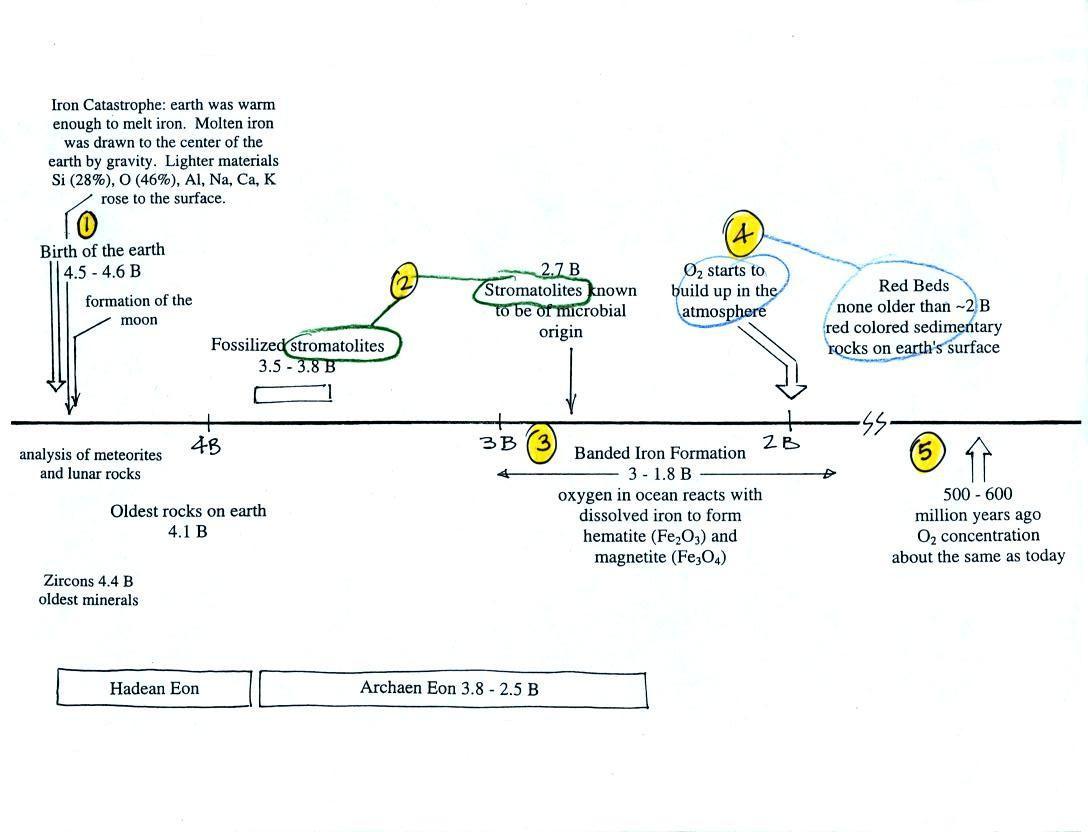
Stromatolites, banded iron, red beds - geological
evidence of oxygen on earth
The
following figure is the first page in the packet of
photocopied ClassNotes.
This somewhat confusing figure shows some of the important
events in the history of the earth and evolution of the
atmosphere. There were 5 main points I wanted you to take
from this figure, and really 1-3 are the most important.
First, Point 1: the
earth is thought to be between 4.5 and 4.6 billion years
old. If you want to remember the earth is a few billion
years old that is probably close enough. A relatively
minor point shown in the figure: the formation of the earth's
molten iron core was important because it gave the earth a
magnetic field. The magnetic field deflects the solar wind
and prevents the solar wind from blowing away our present day
atmosphere.
Stromatolites (Point
2) are geological features, column-shaped structures
made up of layers of sedimentary rock, that are
created by microorganisms living at the top of the
stromatolite (I'm not a geologist and I've never actually seen
a stromatolite, so this is all based on photographs and
written descriptions). Fossils of the very small
microbes (cyanobacteria = blue green algae) have been found in
stromatolites as old as 2.7 B years and are some of the
earliest records of life on earth. Much older (3.5 to
3.8 B year old) stromatolites presumably also produced by
microbes, but without microbe fossils, have also been
found.
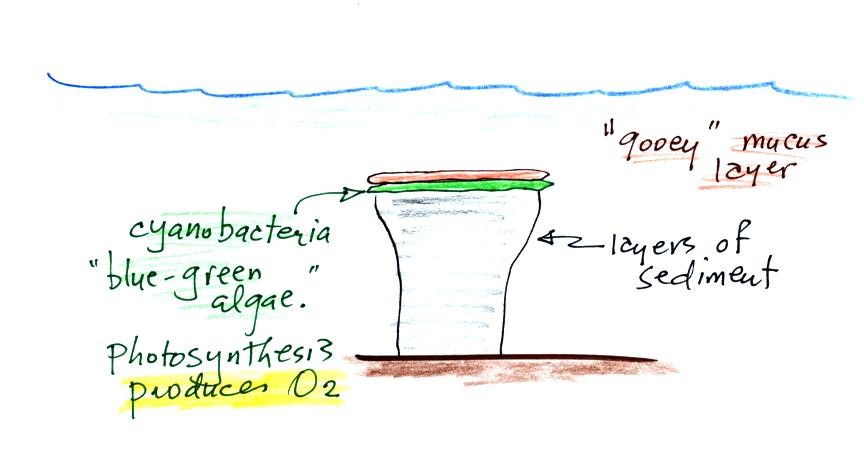
Blue green algae grows at the top of the column, under water
but near the ocean surface where it can absorb sunlight. As
sediments begin to settle and accumulate on top of the algae they
start to block the sunlight. The cyanobacteria would then
move to the top of this sediment layer and the process would
repeat itself. In this way the stromatolite column would
grow layer by layer over time. You might be wondering why we
are learning about stromatolites. It's because the
cyanobacteria on them were able to produce oxygen using
photosynthesis.

|

|
Living stromatolites are found in
a few locations today.The two pictures above are from Lake
Thetis (left) and Shark
Bay (right) in Western Australia (the two
photos above and the photograph below come from this source).
The picture was probably taken at low tide, the stromatolites
would normally be covered with ocean water. It doesn't look
like a good place to go swimming, I would expect the top surfaces
of these stromatolites to be slimy. Hamelin Pool in Western
Australia is a World Heritage Area, the stromatolites there are
the oldest and largest living fossils on earth (see this
source for more information)
Living stromatolites at Highborne Cay in the Bahamas.
Point 3 refers
to the banded iron formation, a type of rock formation.
These rocks are 2 - 3 billion years old (maybe older) and are
evidence of oxygen being produced in the earth's oceans.
Here are a couple of pictures of samples of banded iron formation
rock that I passed around in class.
The main
thing to notice are the alternating bands of red and
black. The rocks are also relatively heavy
because they contain a lot of iron. The next
paragraph and figure explain how these rocks formed.
Banded iron formation
Rain would first of all wash iron ions from the
earth's land surface into the ocean (this was at a
time before there was any oxygen in the
atmosphere). Once in the ocean, the iron ions
reacted with oxygen from the cyanobacteria living in
the ocean water to form hematite or magnetite.
These two minerals precipitated out of the water to
form a layer on the sea bed. This is what
produced the black layers.
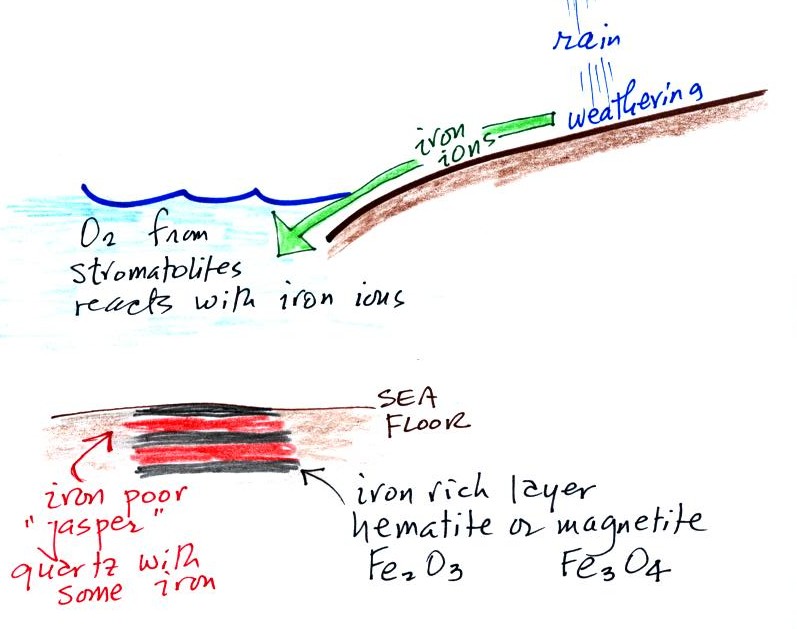
Periodically the oxygen production would
decrease or stop (rising oxygen levels might have killed the
cyanobacteria or seasonal changes in incoming sunlight might
have slowed the photosynthesis). During these times of low
oxygen concentration, red layers of jasper would form on the
ocean bottom. The jasper doesn't contain as much
iron.
Eventually the cyanobacteria would recover, would begin
producing oxygen again, and a new layer of hematite or magnetite
would form. The rocks that resulted, containing
alternating layers of black hematite or magnetite and red layers
of jasper are known as the banded iron formation. In addition to the red and black layers,
you see yellow layers made of fibers of quartz in the samples
passed around class.
Eventually the oxygen in the oceans reacted with all of the
iron ions in the water. Oxygen was then free to diffuse from the ocean
into the atmosphere. Once in the air, the oxygen could react
with iron in sediments on the earth's surface. This produced
red colored (rust colored) sedimentary rock. These are
called "Red Beds" (Point 4).
None of these so-called red beds are older than about 2 B years
old. Thus it appears that a real buildup up of oxygen in the
atmosphere began around 2 B years ago.
Red State Park near Sedona
Arizona. An example of "red beds" that formed during the
Permian period 250-300 million years
ago.
Oxygen concentrations reached levels that are about the same as
today around 500 to 600 million years ago (Point 5 in the figure).