Often, the Earth's atmosphere is divided into several different layers that are defined according to the typical change in air temperature. (See Image Below).
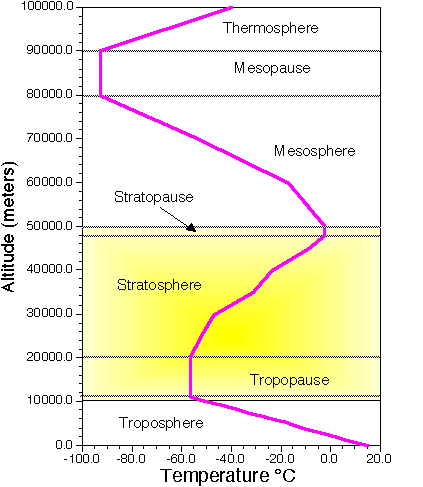 |
| Atmospheric layers defined by structure of air temperature. |
Air density can be defined as the number of air molecules per unit volume (number density). Near sea level there are about 2.7x1019 molecules per cm3(cubic centimeter) or 4.4x1020 molecules per inch3(cubic inch). Air molecules are held near the earth by gravity. In other words, air has weight. Weigh an empty bag, then fill it with air, it now weighs more. In addition gases, like air, are easily compressed, i.e., squeeze a gas together and its number density increases. In other words, we say gases are compressible because they can easily be squeezed into a smaller volume. Solids and liquids on the other hand are not easily compressed.
The weight of all of the air above a given point in the atmosphere squeezes air molecules closer together, which causes their numbers in a given volume to increase (increase in number density). The more air above a level (and hence the more weight of air above a level), the greater the squeezing effect (or compression).
Since air density is the number of air molecules in a given space (volume), air density is typically greatest at the surface or sea level (where it is squeezed by the weight of the entire atmosphere above) and decreases as we move up in the atmosphere because the weight of air above becomes less and hence there is less of a squeezing effect (See Figure Z).
Atmospheric air pressure results from the Earth's gravitational pull on the overlying air. Without gravity holding the atmosphere just above the ground surface, air molecules would spread out, and the gas pressure would be close to zero.
The weight of the atmosphere acts as a force upon the underlying surface of the Earth. The amount of force excerted over an area of surface is called atmospheric pressure or air pressure. Near sea level, the average air pressure is about 14.7 pounds per square inch. In this class we will use the unit millibars(mb) to specify air pressure. At sea level the average air pressure is 1013 mb. Another way to think of this is that the total weight of all the air above sea levels weighs enough to cause 1013 mb of air pressure.
Since the air (a gas) is a fluid, the pressure force acts in all directions, not just downward. The pressure force pushing downward due to the weight of the air is the same as the pressure force acting sideways and even upward. If you are having trouble understanding this, make an analogy with another fluid liquid water. Consider a deep swimming pool full of water. The water pressure anywhere in the pool depends on the weight of the water above (that is the deeper you dive downward in the pool, the stronger the water pressure.) The pressure force is not just downward though, it pushes in on your body from all directions. The average air pressure at sea level (1013 mb or sometimes called one atmosphere of pressure) is caused by the weight of all the air above sea level. In the same way water pressure is caused by the weight of water above you. At a depth of 32 feet (9.75 meters) below a water surface, the water pressure is about one atmosphere. Thus, the entire column of air from sea level to outer space weighs as much as a 32 foot column of water. Of course diving deeper into water means you will encouter an increasing water pressure (enough to crush you if you go too deep).
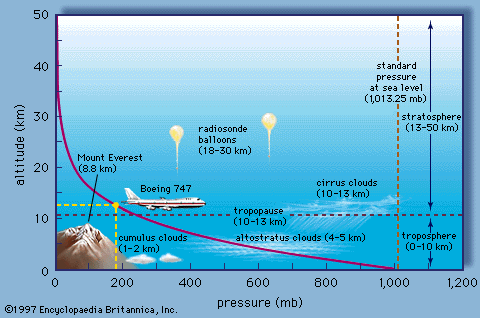 |
| Typical change in air pressure with altitude. Note how rapidly air pressure falls with increasing altitude. |
In the atmosphere, the air pressure at any point caused by the weight per area of the air above that point. As we climb in elevation, fewer air molecules are above us (less weight of air above us); hence, atmospheric pressure always decreases as you move upward in the atmosphere (See Figure B). Another way to look at it is that the air pressure at any point in the atmosphere is exactly enough to support the weight of the column of air above it. A balance exists between the gravitational force pushing air downward and the upward directed pressure force. This balance is called hydrostatic balance (see figure).
Earlier we made an analogy between diving down in water and moving downward in the atmosphere. In both cases, the fluid presure increases as you move down because there is more and more weight of fluid above you. A big difference between water and air, though, is that air is compressible and water is not. This affects the rate of pressure changes as one moves up or down in the fluid as shown in Figure C, which compares the rate of vertical pressure changes between water and air.