Writing is an important part of
this class and is described in more detail on the Writing
Requirements
handout. Please read
through the information on the writing
requirements handout and let
me know if you have any questions.
The first half of your writing grade is an experiment report. You only need to do one of the experiments, so think about which of the experiments listed on the handout you would like to do. I'll bring a signup sheet to class on Wednesday and will probably bring some materials Experiment #1 materials on Friday.
The so-called One Side of One Page (1S1P) reports make up the second part of your writing grade. Topics will appear periodically on the class webpage during the semester. As you write reports you will earn points (the exact number of points will depend on the topic). Your objective should be to earn 45 1S1P pts, the maximum number allowed.
Your grade in this class will depend on your quiz scores, how much extra credit you earn (from optional take home and in class assignments), your writing grade, and (perhaps) your score on the final exam. A sample grade report from the S;pring 2012 class is shown below (the numbers are class averages)
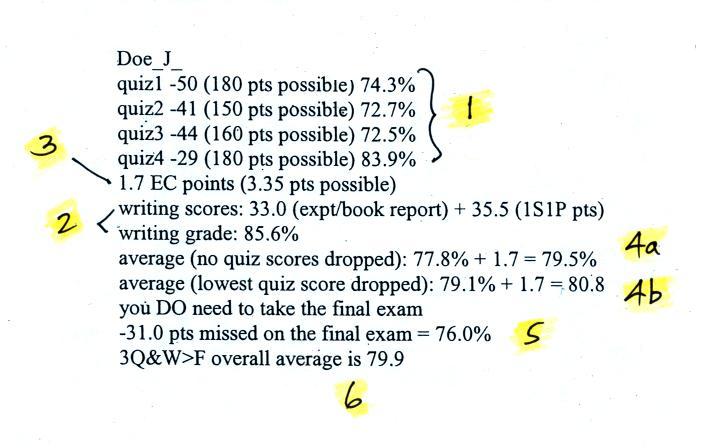
Point 4a shows the average of the 4 quizzes and the writing grade with the extra credit points added in. If this is 90.0% or above you don't have to take the final. If that doesn't happen you will have to take the final exam but the average will be recomputed with the lowest quiz score dropped. That is shown at Point 4b.
If you do well on the final exam it will count 40% of your overall grade. If you don't do so well on the final it only counts 20%. In this example the final exam score (76%) was lower than the semester average (80.8%) so the final only counted 20%. The overall score ended up 79.9. I would probably round this up to 80% and give this student a B. Note that if the student had earned just an additional 0.1 pts of extra credit or a few more 1S1P pts a B grade would have been assured.
We did cover a little course material in class today just so you can get an idea of how that will work. If we were using a book we'd start in Chapter 1 and here's some of what we would be looking at first in this course.

Can you see air?
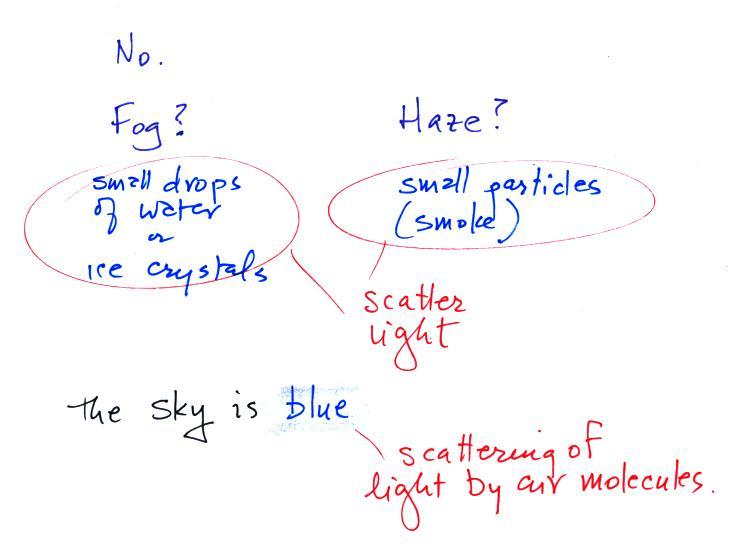
This is a little
more detailed
answer to this question than was given in class. Air is
normally clear, transparent, and invisible (that would be true of the
air in the
classroom). Sometimes the air looks
foggy, hazy or smoggy. In these cases you are seeing the effects
of
small water droplets or ice crystals (fog) or small particles of dust
or smoke (haze and smog). The particles themselves may be too
small
to be seen with
the naked eye but are visible because they scatter (redirect)
light. Scattering is a pretty important concept and we will learn
more about it in a week
or so.
The atmosphere isn't actually clear. When sunlight shines through the atmosphere the sky appears blue. This is a little more complicated form of scattering of sunlight by air molecules. We'll come back to this later as well.
The atmosphere isn't actually clear. When sunlight shines through the atmosphere the sky appears blue. This is a little more complicated form of scattering of sunlight by air molecules. We'll come back to this later as well.
Can you smell air?

Natural gas (methane) used in hot water heaters, some stoves, and furnaces is odorless. A chemical (mercaptan) is added to natural gas so that you can smell it and know when there is a leak before it builds up a concentration that could cause an explosion.
Can you feel air

It
is harder to answer this question. We're always in contact with
air. Maybe we've grown so accustomed to it we aren't aware of how
it feels. We can certainly feel whether
the air is hot or cold, but that have more to do with energy exchange
between us and our surroundings. And we can feel wind.
We will see that here in the classroom air pressure is pressing on every square inch of our bodies with 12 or 13 pounds of force. If that were to change suddenly I'm pretty sure we'd feel it and it would probably really hurt.
What are the 5 most abundant gases in air?
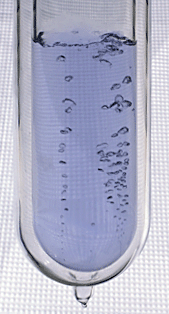
Let's start with the most abundant gas in the atmosphere. I poured some of this same material (in liquid form) into a styrofoam cup. Here's a photo I took back in my office.

You can see the liquid, it's clear, it looks like water. At least one student (probably many more) knew that this was liquid nitrogen.
The most abundant gas in the atmosphere is nitrogen. We'll use liquid nitrogen in several class demonstration this semester.
Nitrogen was discovered in 1772 by Daniel Rutherford (a Scottish botanist). Atmospheric nitrogen is relatively unreactive and is sometimes used to replace air in packaged foods to preserve freshness.
Oxygen is the second most abundant gas in the atmosphere. Oxygen is the most abundant element (by mass) in the earth's crust, in ocean water, and in the human body.
A couple of photographs of liquid oxygen are shown above. It has a (very faint) blue color (I was pretty disappointed when I saw the pictures for the first time because I had imagined the liquid oxygen might be a deep vivid blue).
When heated (such as in an automobile engine) the oxygen and nitrogen in air react to form compounds such as nitric oxide (NO), nitrogen dioxide (NO2), and nitrous oxide (N2O). Together as a group these are called oxides of nitrogen; the first two are air pollutants, the last is a greenhouse gas.
We will see that here in the classroom air pressure is pressing on every square inch of our bodies with 12 or 13 pounds of force. If that were to change suddenly I'm pretty sure we'd feel it and it would probably really hurt.
What are the 5 most abundant gases in air?
Let's start with the most abundant gas in the atmosphere. I poured some of this same material (in liquid form) into a styrofoam cup. Here's a photo I took back in my office.

You can see the liquid, it's clear, it looks like water. At least one student (probably many more) knew that this was liquid nitrogen.
The most abundant gas in the atmosphere is nitrogen. We'll use liquid nitrogen in several class demonstration this semester.
Nitrogen was discovered in 1772 by Daniel Rutherford (a Scottish botanist). Atmospheric nitrogen is relatively unreactive and is sometimes used to replace air in packaged foods to preserve freshness.
Oxygen is the second most abundant gas in the atmosphere. Oxygen is the most abundant element (by mass) in the earth's crust, in ocean water, and in the human body.
 |
 |
| from: http://www.webelements.com/oxygen/ The webelements site credits Prof. James Marshall's Walking Tour of the Elements. |
from: http://en.wikipedia.org/wiki/Oxygen Wikipedia credits Dr. Warwick Hillier of Australia National University |
A couple of photographs of liquid oxygen are shown above. It has a (very faint) blue color (I was pretty disappointed when I saw the pictures for the first time because I had imagined the liquid oxygen might be a deep vivid blue).
When heated (such as in an automobile engine) the oxygen and nitrogen in air react to form compounds such as nitric oxide (NO), nitrogen dioxide (NO2), and nitrous oxide (N2O). Together as a group these are called oxides of nitrogen; the first two are air pollutants, the last is a greenhouse gas.
Here are the 5 most abundant gases
in the earth's atmosphere.

Water vapor and argon are the 3rd and 4th most abundant gases in the atmosphere. A 2% water vapor concentration is listed above but it can vary from near 0% to as high as 3% or 4%. Water vapor is, in many locations, the 3rd most abundant gas in air. In Tucson most of the year, the air is dry enough that argon is in 3rd position and water vapor is 4th.
Water vapor, a gas, is invisible. Clouds are visible because they are made up of small drops of liquid water or ice crystals. We can see clouds even though the individual water droplets are too small to be seen because they scatter light. Water is the only compound that exists naturally in solid, liquid, and gaseous phases in the atmosphere.
Argon is an unreactive noble gas (helium, neon, krypton, xenon, and radon are also inert gases).
Here's a little more explanation (from Wikipedia) of why noble gases are so unreactive. Don't worry about all these additional details. The noble gases have full valence electron shells. Valence electrons are the outermost electrons of an atom and are normally the only electrons that participate in chemical bonding. Atoms with full valence electron shells are extremely stable and therefore do not tend to form chemical bonds and have little tendency to gain or lose electrons.
 |
 |
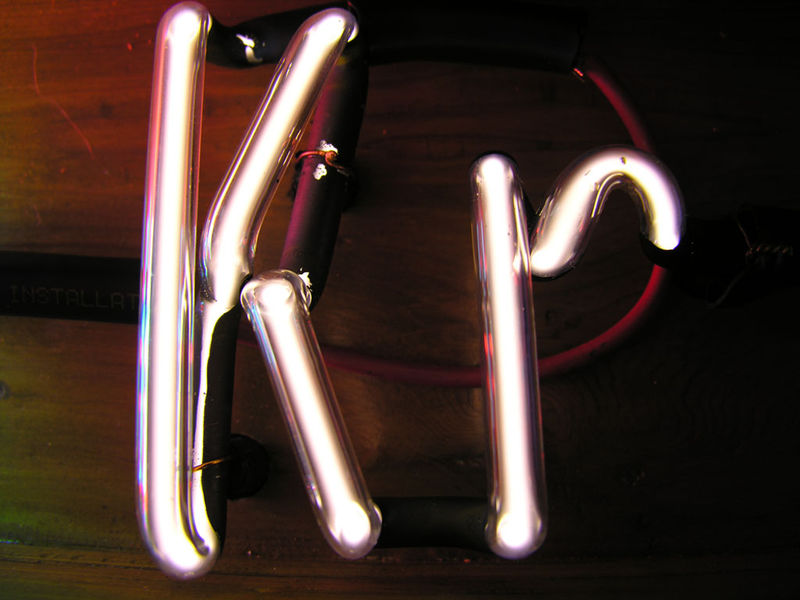 |
 |
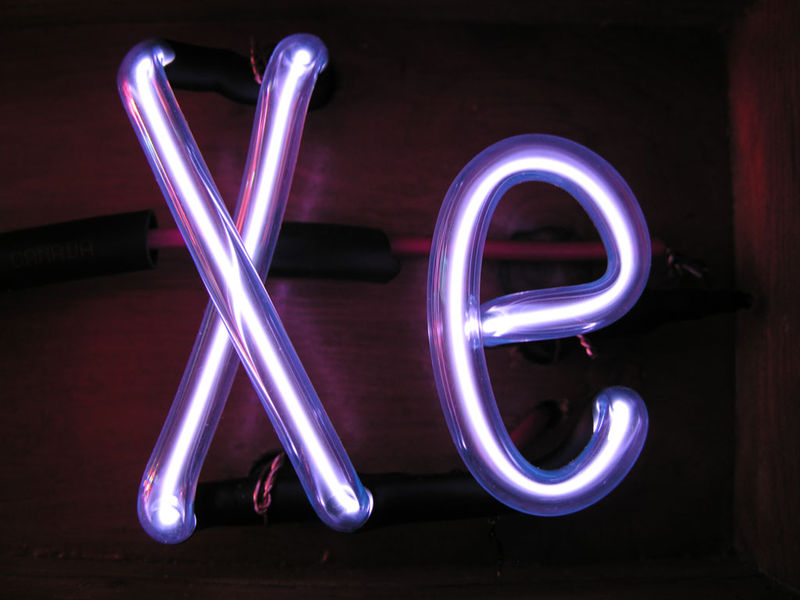 |
Noble gases are often used used in neon signs; argon produces a blue color. The colors produced by Argon (Ar), Helium (He), Kryton (Kr), Neon (Ne) and Xenon (Xe), which are also noble gases, are shown above (source of the images). This picture wasn't shown in class.
This is about as far as we got in class on Monday. Here's a little more information to finish off this topic. We'll go over this again at the start of class on Wednesday.
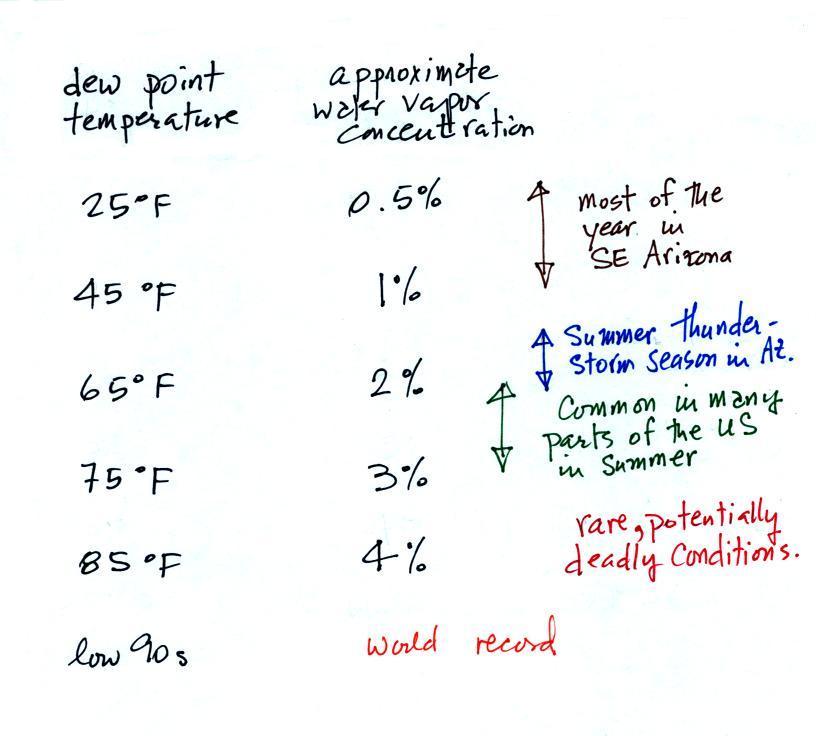
Water plays an important role in the formation of clouds, storms, and weather. Meteorologists are very interested in knowing and keeping track of how much water vapor is in the air at a particular place and time. One of the variables they use is the dew point temperature. The value of the dew point gives you an idea of how much water vapor is actually in the air. The higher the dew point value, the more water vapor the higher the water vapor concentration.