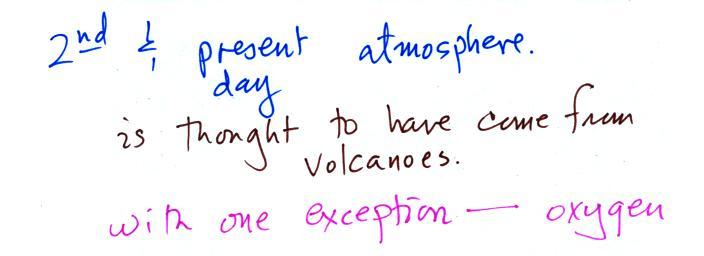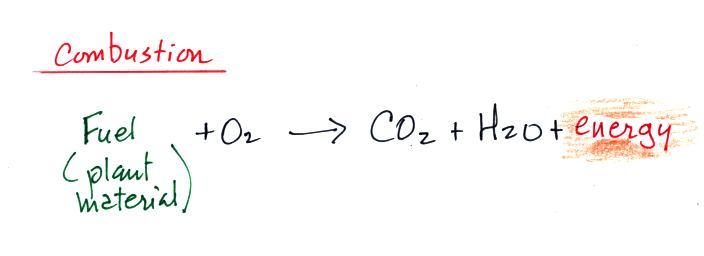Don't worry
about remembering all of the gases listed above. Volcanoes emit a
lot of
water vapor and carbon
dioxide. As
the earth began to cool the water vapor condensed and began to create
and fill oceans. Carbon dioxide dissolved in the oceans and was
slowly
turned into rock. Smaller amounts of nitrogen (N2) are emitted by
volcanoes. Nitrogen is relatively unreactive and remained in the
air. Nitrogen concentration built up over time. We'll learn
a little more about sulfur dioxide when we cover air pollutants.
Volcanoes didn't add any of the oxygen that is today's
atmosphere.
Where did that come from?
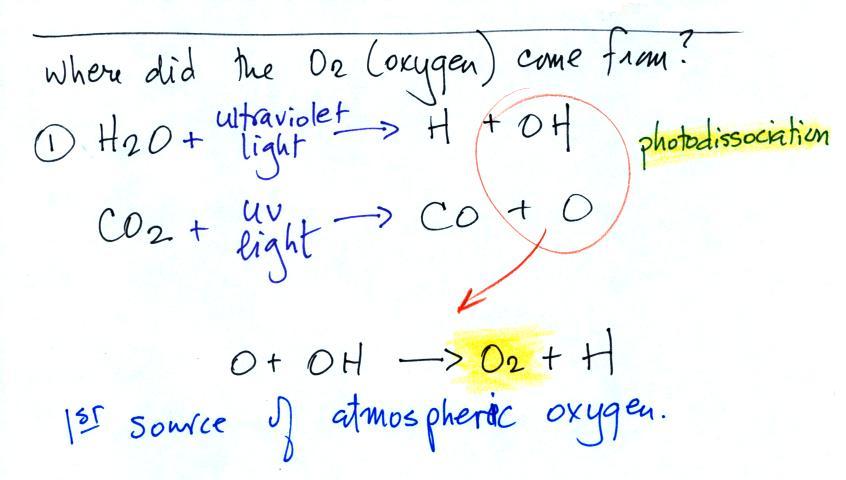
The oxygen is thought to have first
come from
photodissociation of
water vapor and carbon dioxide by ultraviolet light (the high energy
UV light is able to split the H20
and CO2
molecules into
pieces). The O and OH then react
to form O2 and H.
By the way I don't expect you to remember
the chemical formulas in the example above. It's often easier and
clearer to show what is happening in a chemical formula than to write
it out in words. If I were to right the equations down, you
should be able to interpret them. It's probably also good to
remember that
ultraviolet light is capable of breaking molecules apart (I think
you'll see the dissociation reactions enough that you won't have any
trouble remembering them).
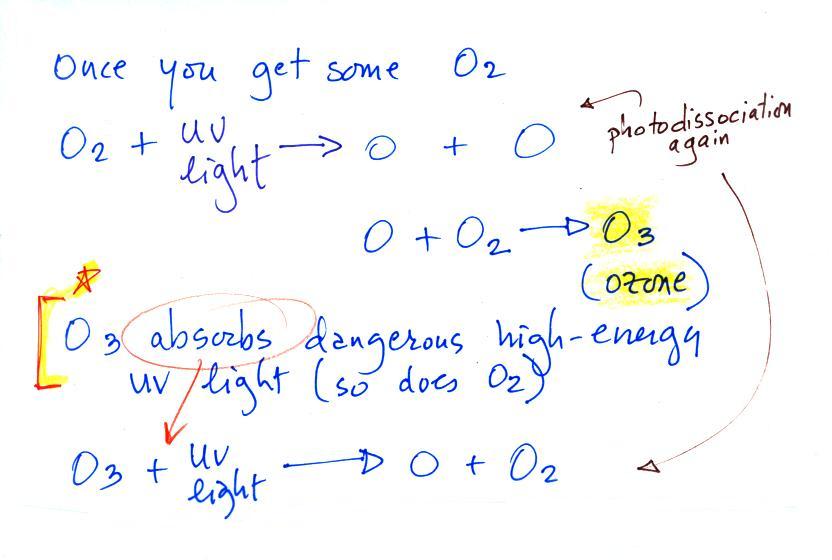
Once molecular oxygen (O2) begins to accumulate in the
air UV light can split it apart to make atomic oxygen (O). The
atoms of oxygen can react with molecular oxygen to form
ozone (O3).
Ozone in the atmosphere began to absorb ultraviolet
light and life forms could safely move from the oceans (which offered
protection from UV light in the
absence of ozone) onto land.
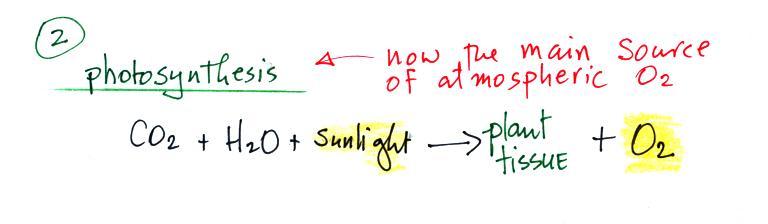
Photosynthesis is the 2nd
(photodissociation of CO2
and H2O
was the 1st) and now the main source of atmospheric oxygen.
Note below that combustion is really just the opposite of
photosynthesis.
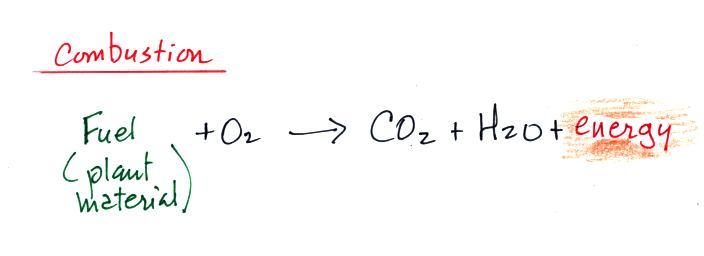
We burn fossil fuels (dead,
undecayed plant material) to generate energy.
Water vapor and carbon dioxide are by products. Combustion is a
source of CO2.
We'll see these two equations again when we study the greenhouse
effect (CO2 is
a
greenhouse
gas
) and global warming.
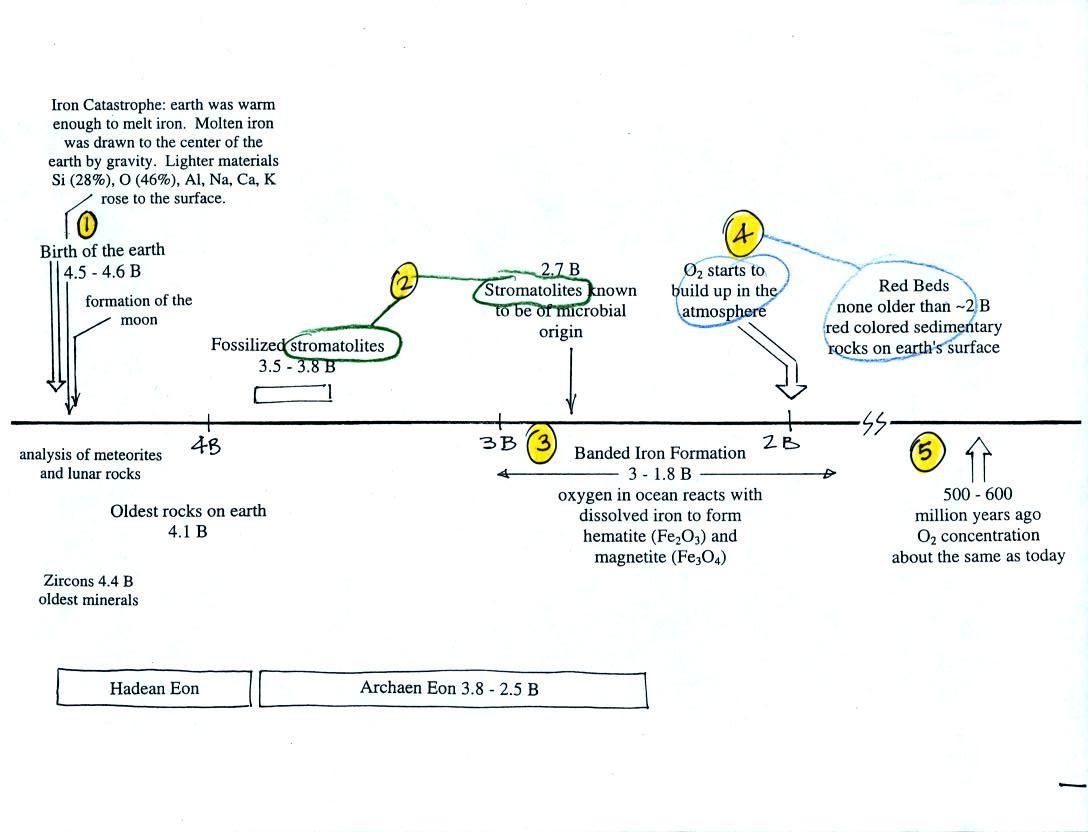
We had a quick look at the following figure (p. 1 in the packet of
photocopied
ClassNotes) which finished up this section on the origin and evolution
of the earth's atmosphere.
This somewhat confusing
figure shows some of the important events in the history of the earth
and evolution of the atmosphere. The numbered points were
emphasized.
First, Point 1: the earth
is thought to be between 4.5
and 4.6 billion years old. If you want to remember the earth is a
few
billion years old that is probably close enough.
The iron catastrophe was an important event (but wasn't
discussed in class). Circulation of liquid metal in the
core of
the earth gives the earth a magnetic field. The magnetic field
deflects the solar wind around the earth. Remember the solar wind
may have swept away the earth's original atmosphere.
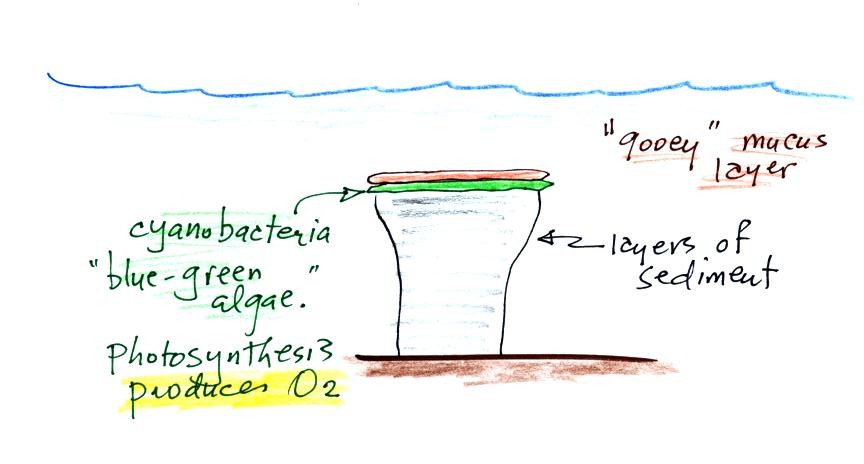
Stromatolites
(Point
2)
are
column-shaped
structures
made
up of layers of sedimentary rock. They are created by
microorganisms
living at the top of the stromatolite (I've never actually seen a
stromatolite, so this is all based on photographs and written
descriptions). Fossils of the very small microbes (cyanobacteria
= blue green algae)
have been found in stromatolites as old as 2.7 B years and are some of
the earliest records of life on earth. Much older (3.5 to 3.8
B years old) stromatolites, presumably also produced by microbes, but
without
microbe fossils, have been found.
We're learning about stromatolites
because the cyanobacteria were able to produce oxygen using
photosynthesis.
Living stromatolites are found
in a
few locations today. The picture above is from Coral Bay Australia, located on
the
western tip of the continent. The picture was probably taken at
low tide, the stromatolites would normally be covered with ocean water.
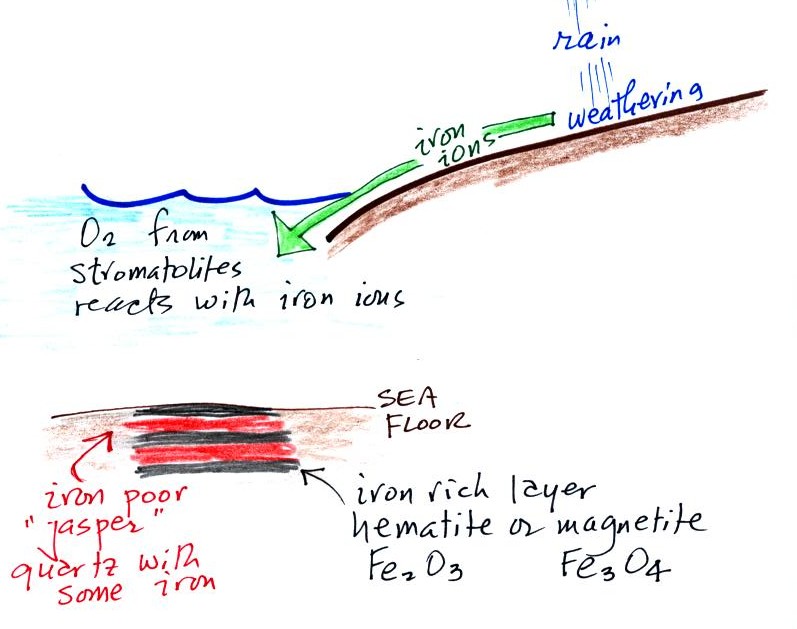
Once cyanobacteria began to produce
oxygen in ocean water, the oxygen reacted with dissolved iron (iron
ions in the figure below) to form hematite or magnetite. These
two minerals precipitated out of the water to form a layer on the sea
bed.